Mosquito- and tick-borne orthoflaviviruses cross an in vitro endothelial-astrocyte barrier
- PMID: 40673004
- PMCID: PMC12263646
- DOI: 10.3389/fcimb.2025.1624636
Mosquito- and tick-borne orthoflaviviruses cross an in vitro endothelial-astrocyte barrier
Abstract
Introduction: The genus Orthoflavivirus of the Flaviviridae family includes several notable pathogens such as mosquito-borne West-Nile virus (Orthoflavivirus nilense, WNV) and Tick-borne encephalitis virus (Orthoflavivirus encephalitidis, TBEV) that are highly neurotropic and may cause severe neurological disease leading to lifelong disabilities, coma and death. These viruses have developed mechanisms to breach the compact blood-brain barrier (BBB) and establish infection within the central nervous system (CNS). Nevertheless, neuroinvasive mechanisms of orthoflaviviruses remain poorly understood. Complex anatomy of the CNS and the organization of the BBB is a major challenge to study neuroinvasion of orthoflaviviruses in vivo. Therefore, in vitro BBB models are useful tools to study direct interaction of viruses with the endothelial barrier.
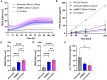
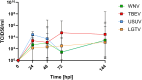
Methods: In this study, we employed an in vitro transwell BBB model comprising primary mouse brain microvascular endothelial cells and astrocytes to compare the ability of mosquito-borne and tick-borne orthoflaviviruses to cross a compact endothelial barrier and reach the basolateral compartment of the transwell system. The influence of virus inoculation on the barrier properties was determined by measuring transendothelial electrical resistance (TEER).
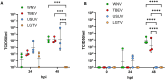
Results: The results demonstrate that while pathogenic WNV and TBEV cross the endothelial barrier the ability of low pathogenic Usutu virus (USUV) and Langat virus (LGTV) was inconsistent. All viruses tested display virus replication within the endothelial cells. Nevertheless, virus replication did not affect the barrier function of endothelial cells as demonstrated by sustained TEER and absence of leakage of high molecular weight dextran molecules through the endothelial barrier even at several hours post infection.
Discussion: Our findings indicate that orthoflaviviruses can infect the endothelial cells, replicate within them without affecting the cells and its barrier function. Nevertheless, only pathogenic WNV and TBEV showed the ability to cross the endothelial barrier and reach the basolateral compartment.
Keywords: astrocytes; blood-brain barrier; endothelial cells; neuroinvasion; orthoflavivirus; transendothelial electrical resistance.
Copyright © 2025 Schweitzer, Schröder, Friedrichs, Gudi, Skripuletz, Steffen, Palus, Růžek, Osterhaus and Prajeeth.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

References
-
- Agliani G., Giglia G., Marshall E. M., Gröne A., Rockx B. H. G., van den Brand J. M. A. (2023). Pathological features of West Nile and Usutu virus natural infections in wild and domestic animals and in humans: A comparative review. One Health 16, 100525. doi: 10.1016/j.onehlt.2023.100525 - DOI - PMC - PubMed
-
- da Fonseca N. J., Lima Afonso M. Q., Pedersolli N. G., de Oliveira L. C., Andrade D. S., Bleicher L. (2017). Sequence, structure and function relationships in flaviviruses as assessed by evolutive aspects of its conserved non-structural protein domains. Biochem. Biophys. Res. Commun. 492, 565–571. doi: 10.1016/j.bbrc.2017.01.041 - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

