Comparative transcriptome analysis of genes related to tuberization in late and early maturing potato (Solanum tuberosum) cultivars
- PMID: 40676087
- PMCID: PMC12271483
- DOI: 10.1038/s41598-025-11875-1
Comparative transcriptome analysis of genes related to tuberization in late and early maturing potato (Solanum tuberosum) cultivars
Abstract
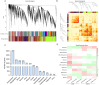
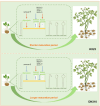
Potato maturity is a complex and vital agronomic trait directly impacting yield, quality, and economic value. Despite its importance, the molecular regulatory networks governing potato maturity remain largely unexplored. In this study, the factors underlying maturity differences between early-maturing (KX23) and late-maturing (DN310) tetraploid potato cultivars were investigated for the first time under field conditions. Tuber yields, starch content, flowering time, maturity and the number of morphologically modified stolons and initiated tubers were measured, revealing that maturity differences between KX23 and DN310 are linked to the timing of tuberization processes. To explore the molecular basis of these differences, RNA-seq analysis was performed on hooked stolons, swollen stolons, and initiated tubers, identifying key pathways involved in tuberization. WGCNA and qRT-PCR further pinpointed critical genes contributing to these pathways. DEGs between the two cultivars were primarily enriched in secondary metabolite pathways. Notably, a significant overlap of DEGs between KX23 and DN310 across the three developmental stages was identified, with enrichment in carbohydrate metabolism pathways. In total, 28 candidate genes consistently involved in tuber induction and formation were identified. Based on their functions, a model explaining how KX23 achieves faster tuberization, shortening its maturity period, was proposed. These findings provide valuable insights into the molecular regulatory mechanisms of tuberization and maturity in potatoes.
Keywords: Maturity; Potato; RNA-seq; Tuberization; WGCNA.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures






Similar articles
-
Gibberellin 2-Oxidases in Potato (Solanum tuberosum L.): Cloning, Characterization, In Silico Analysis and Molecular Docking.Mol Biotechnol. 2024 Apr;66(4):902-917. doi: 10.1007/s12033-023-00745-8. Epub 2023 Apr 16. Mol Biotechnol. 2024. PMID: 37061992
-
The potential role of short internodes/stylish (SHI/STY) and SHI related sequence (SRS) in potato tuberization.BMC Plant Biol. 2025 Aug 25;25(1):1124. doi: 10.1186/s12870-025-07172-8. BMC Plant Biol. 2025. PMID: 40855269 Free PMC article.
-
StMYB308L and its regulatory role in flavonoid biosynthesis pathways in potato.BMC Plant Biol. 2025 Jul 8;25(1):890. doi: 10.1186/s12870-025-06897-w. BMC Plant Biol. 2025. PMID: 40629300 Free PMC article.
-
Molecular signals that govern tuber development in potato.Int J Dev Biol. 2020;64(1-2-3):133-140. doi: 10.1387/ijdb.190132ab. Int J Dev Biol. 2020. PMID: 32659001 Review.
-
Health professionals' experience of teamwork education in acute hospital settings: a systematic review of qualitative literature.JBI Database System Rev Implement Rep. 2016 Apr;14(4):96-137. doi: 10.11124/JBISRIR-2016-1843. JBI Database System Rev Implement Rep. 2016. PMID: 27532314
References
-
- Camire, M. E., Kubow, S. & Donnelly, D. J. Potatoes and human health. Crit. Rev. Food Sci. Nutr.49, 823–840. 10.1080/10408390903041996 (2009). - PubMed
-
- Jennings, S. A. et al. Global potato yields increase under climate change with adaptation and CO2 fertilisation. Front. Sustainable Food Syst.410.3389/fsufs.2020.519324 (2020).
-
- Birch, P. R. J. et al. Crops that feed the world 8: potato: are the trends of increased global production sustainable? Food Secur.4, 477–508. 10.1007/s12571-012-0220-1 (2012).
-
- Kawar, P. G. et al. In achieving sustainable cultivation of potatoes 1 burleigh Dodds series in. Agric. Sci. 143–168 (2018).
-
- He, M. et al. Comprehensive transcriptome profiling and transcription factor identification in early/late leaf senescence grafts in potato. Physiol. Plant.176, e14582. 10.1111/ppl.14582 (2024). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- GA23B011/Heilongjiang Province key research and development plan
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CX23GG02/Heilongjiang Province agricultural science and technology innovation span project
- CARS-09/Agriculture Research System of China
- CARS-09/Agriculture Research System of China
- CARS-09/Agriculture Research System of China
- CARS-09/Agriculture Research System of China
- CARS-09/Agriculture Research System of China
- CZKYF2024-1-C024/Research Business Expenses Project of Heilongjiang Province's Provincial Research Institutes
- CZKYF2024-1-C024/Research Business Expenses Project of Heilongjiang Province's Provincial Research Institutes
- CZKYF2024-1-A005/Heilongjiang provincial institute research expenses project
- CZKYF2024-1-A005/Heilongjiang provincial institute research expenses project
LinkOut - more resources
Full Text Sources

