Curdione protects vascular endothelial cells and atherosclerosis via the regulation of DNMT1-mediated ERBB4 promoter methylation
- PMID: 40677584
- PMCID: PMC12269948
- DOI: 10.1515/med-2025-1223
Curdione protects vascular endothelial cells and atherosclerosis via the regulation of DNMT1-mediated ERBB4 promoter methylation
Abstract
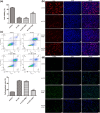
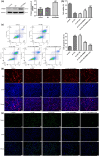
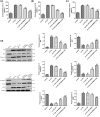
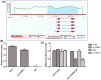
Atherosclerosis (AS) is initiated by the activation of the endothelial cells, which is followed by a series of events that trigger the narrowing of blood vessels and the activation of inflammation. This study aimed to investigate in vitro the roles and underlying mechanisms of curdione in AS. Human umbilical vein endothelial cells (HUVECs) were stimulated with oxidized low-density lipoprotein (ox-LDL) and then treated with curdione, after which the growth of the HUVECs and the related mechanisms were determined. HUVECs with ERBB4 overexpression were constructed to explore the role of ERBB4 in curdione-mediated AS. The interaction among ERBB4, methylation, and curdione was confirmed by chromatin immunoprecipitation (ChIP)-quantitative PCR (qPCR) and dual luciferase reporter gene assays. Both curdione and ERBB4 overexpression individually and significantly enhanced viability and proliferation while suppressing apoptosis of the ox-LDL-induced HUVECs, and the combination of curdione and ERBB4 overexpression had better effects. Compared with the ox-LDL-induced HUVECs, both curdione and ERBB4 overexpression individually decreased the levels of IL-6, IL-1β, and IL-8 (P < 0.05). They also upregulated Bax, caspase-3, E-cadherin, and F-actin while downregulating Bcl-2 and VEGF (P < 0.05). Additionally, the ERBB4 bound to the DNMT1 gene, and the curdione participated in AS via the ERBB4 gene. The study demonstrated that either curdione or ERBB4 overexpression individually may ameliorate AS development by inhibiting apoptosis, inflammation, and the EndMT of HUVECs. In addition, curdione may protect the vascular endothelial cells and AS by regulating the DNMT1-mediated ERBB4 promoter methylation.
Keywords: DNMT1; ERBB4; HUVECs; atherosclerosis; curdione; ox-LDL.
© 2025 the author(s), published by De Gruyter.
Conflict of interest statement
Conflict of interest: The authors state no conflict interest.
Figures

References
-
- El Khoudary SR, Aggarwal B, Beckie TM, Hodis HN, Johnson AE, Langer RD, et al. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation. 2020;142(25):e506–32. 10.1161/cir.0000000000000912. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
