Selective blockade of acid-sensing ion channel 1a can provide substantial hippocampal neuroprotection
- PMID: 40677921
- PMCID: PMC12267221
- DOI: 10.3389/fcell.2025.1582970
Selective blockade of acid-sensing ion channel 1a can provide substantial hippocampal neuroprotection
Abstract
Background: Acid-sensing ion channel 1a (ASIC1a) is the only member of the ASIC family where Ca2+ osmosis has been reported, and it is highly expressed in neurons of the central nervous system. This study aimed to investigate whether ASIC1a is trafficked to the plasma membrane and regulated by the Rho/ROCK and PI3K signaling pathways in temporal lobe epilepsy (TLE). In addition, further research is required to determine whether selective ASIC1a blockade is a viable therapeutic strategy for TLE.
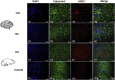
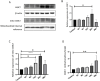
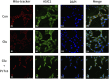
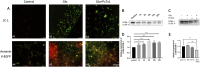
Methods: The localization and expression levels of ASIC1 and mRNA levels of ASIC1a were detected when the Rho/ROCK and PI3K signaling pathways were activated and inhibited in glutamate (Glu)-induced cell. Meanwhile, we analyzed the location and expression of ASIC1 using Western blotting and immunofluorescence in brain tissue samples from TLE patients, kainic acid (KA)-treated rats, and Glu-induced primary hippocampal neurons. Currently, no specific ASIC1a antibody is available, so the ASIC1 antibody was used in this study, as in previous studies. Furthermore, we evaluated the HT22 cell survival rate, mitochondrial damage, apoptosis, and autophagy to examine whether selective blocking ASIC1a (PcTx1) could play a neuroprotective role.
Results: First, the Rho/ROCK and PI3K signaling pathways affect the regulation of the expression and localization of ASIC1, especially the mRNA levels of ASIC1a in the Glu-induced HT22 cell injury model. Second, the high expression of ASIC1 in epilepsy patients was verified in all three sample types, and the phenomenon of its transport from the cytoplasm to the cell membrane/mitochondria was confirmed. Finally, although ASIC1 has a limited epileptogenic effect in the acute phase of epilepsy in vivo, selective blockade of ASIC1a using PcTx1 provided significant hippocampal neuroprotection and reduced mitochondrial damage, apoptosis, and cellular autophagy in vitro.
Interpretation: This study is a systematic report concerning ASIC1a in temporal lobe epilepsy, including in vivo and in vitro experiments addressing both the acute and chronic phases. It provides foundational research for proposing ASIC1a as a new target for epilepsy treatment.
Keywords: ASIC1a; PI3K; Rho/ROCK; hippocampus; neuroprotection; temporal lobe epilepsy.
Copyright © 2025 Li, Cheng, Ma, Li, Zhao, Jiang and Meng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The reviewer ZL declared a shared affiliation with the authors to the handling editor at the time of review.
Figures



Similar articles
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
[Volume and health outcomes: evidence from systematic reviews and from evaluation of Italian hospital data].Epidemiol Prev. 2013 Mar-Jun;37(2-3 Suppl 2):1-100. Epidemiol Prev. 2013. PMID: 23851286 Italian.
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Antidepressants for pain management in adults with chronic pain: a network meta-analysis.Health Technol Assess. 2024 Oct;28(62):1-155. doi: 10.3310/MKRT2948. Health Technol Assess. 2024. PMID: 39367772 Free PMC article.
References
-
- Ali A., Pillai K. P., Ahmad F. J., Dua Y., Vohora D. (2006). Anticonvulsant effect of amiloride in pentetrazole-induced status epilepticus in mice. Pharmacol. Rep. P. R. 58 (2), 242–245. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

