Identification of genetic variations linked to buparvaquone resistance in Theileria annulata infecting dairy cattle in India
- PMID: 40679975
- PMCID: PMC12273908
- DOI: 10.1371/journal.pone.0326243
Identification of genetic variations linked to buparvaquone resistance in Theileria annulata infecting dairy cattle in India
Abstract
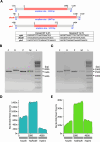
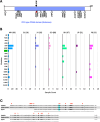
Buparvaquone (BPQ) is used for the treatment of bovine theileriosis, a tickborne disease caused by parasites of the Theileria genus. Studies on T. annulata have linked the mechanism of BPQ resistance predominantly to genetic variations in the parasite cytochrome b (cytb) gene. In addition, cryptic mechanisms of resistance involving the parasite peptidyl-prolyl isomerase (pin1) and dihydroorotate dehydrogenase (dhodh) genes require assessment. In India, where bovine theileriosis is endemic, and BPQ is widely used for treatment, it is necessary to establish the prevalence of genetic variations linked to BPQ resistance. In this study, multiplexed PCR amplification and nanopore sequencing method was used for genotyping the complete gene loci of the three target genes. Analysis of 454 T. annulata field samples collected from seven different states of India revealed the presence of previously reported BPQ resistance associated variations S129G, A146T and P253S in cytb gene and A53P in pin1 gene. The A146T and I203V variations in cytb were found to be prevalent and mostly co-occurring, and their role in BPQ resistance needs further evaluation. This study has revealed the presence of previously reported BPQ resistance-linked mutations in cytb and pin1 genes in T. annulata infecting dairy cattle in India and establishes an Oxford nanopore sequencing method suitable for large-scale surveillance of genetic variation in Theileria parasites from field samples.
Copyright: © 2025 Musale et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Gondard M, Cabezas-Cruz A, Charles RA, Vayssier-Taussat M, Albina E, Moutailler S. Ticks and Tick-Borne Pathogens of the Caribbean: Current Understanding and Future Directions for More Comprehensive Surveillance. Front Cell Infect Microbiol. 2017;7:490. doi: 10.3389/fcimb.2017.00490 - DOI - PMC - PubMed
-
- Zeb J, Song B, Aziz MU, Hussain S, Zarin R, Sparagano O. Diversity and Distribution of Theileria Species and Their Vectors in Ruminants from India, Pakistan and Bangladesh. Diversity. 2022;14(2):82. doi: 10.3390/d14020082 - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

