Melanocyte dysfunctions: future and promise of stem cells
- PMID: 40686746
- PMCID: PMC12267124
- DOI: 10.62347/EOIC7075
Melanocyte dysfunctions: future and promise of stem cells
Abstract
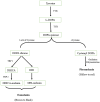
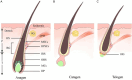
Human melanocytes (MCs) and melanocyte stem cells (McSCs) are integral to skin pigmentation and appendage pigmentation, originating embryonically from neural crest cells. In adult skin, McSCs residing in the epidermis sustain the continuous regeneration of functional melanocytes, a process vital for skin homeostasis and repair. Advances in McSC research have unravelled their pivotal roles in combating disorders such as vitiligo, hair greying, impaired wound healing, and melanoma. Previous studies have significantly advanced our knowledge of the cellular and molecular characteristics of this unique stem cell population. However, a comprehensive understanding of their characteristics in melanocyte dysfunctions leading to conditions like vitiligo is still lacking. Dysfunction or depletion of McSCs is linked to these conditions, highlighting their significance in maintaining skin health. Cutting-edge technologies like single-cell RNA sequencing, spatial transcriptomics, gene editing, and whole-genome sequencing have deepened our understanding of McSC biology and their regulatory microenvironment. This review delves into the latest discoveries, offering a comprehensive perspective on McSCs and their therapeutic potential. By identifying specific molecular signals and crosstalk mechanisms, McSC research opens avenues for regenerative medicine applications, including skin repigmentation, tissue repair, and cancer treatment. The field's progression sets the stage for transformative breakthroughs in skin regeneration and broader regenerative therapies.
Keywords: Melanocyte stem cell; hair greying; homeostasis; melanoma; microenvironment.
AJSC Copyright © 2025.
Conflict of interest statement
None.
Figures

References
-
- Ali SA, Parveen N. The vertebrate pigmentary system: from pigment cells to disorders. Bentham Science Publishers. 2021
-
- Ali SA, Naaz I, Zaidi KU, Ali AS. Recent updates in melanocyte function: the use of promising bioactive compounds for the treatment of hypopigmentary disorders. Mini Rev Med Chem. 2017;17:785–798. - PubMed
-
- Ali SA, Naaz I. Current challenges in understanding the story of skin pigmentation-Bridging the morpho-anatomical and functional aspects of mammalian melanocytes. Muscle Cell and Tissue. 2015:262–85.
Publication types
LinkOut - more resources
Full Text Sources
