Photodynamically tunable ROS-generating hydrogels for accelerated tissue regeneration
- PMID: 40688654
- PMCID: PMC12273516
- DOI: 10.1016/j.bioactmat.2025.05.006
Photodynamically tunable ROS-generating hydrogels for accelerated tissue regeneration
Abstract
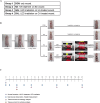
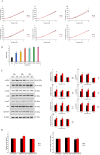
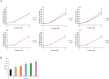
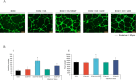
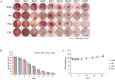
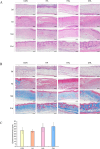
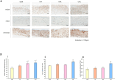
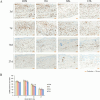
Wound healing progresses through many key cellular activities, including fibroblast and keratinocyte proliferation and angiogenesis. This study explored the wound-healing potential of reactive oxygen species (ROS)-generating hyaluronic acid (HA) hydrogels. We fabricated a chlorin e6-conjugated HA (Ce6-HA) hydrogel that generates ROS when subjected to irradiation from an LED light source. In vitro studies revealed that the ROS generated by the Ce6-HA hydrogels enhanced the proliferation of fibroblasts and keratinocytes. Further, the fibroblasts were found to have high levels of intracellular ROS, elevated expression of p-ERK1/2, p-p38 MAPK, p-Akt, and cyclin D1 proteins, and enhanced collagen deposition. Moreover, the Ce6-HA hydrogel also promoted endothelial angiogenesis in vitro. In vivo studies demonstrated the ROS-generating HA hydrogels significantly improved wound closure and tissue regeneration compared to control groups. The Ce6-HA hydrogel-treated group exhibited accelerated wound healing, with enhanced fibroblast proliferation, increased keratinocyte proliferation, and better angiogenesis. Histopathological and immunohistochemical analyses showed elevated levels of key growth factors and signaling molecules, which are critical to wound healing. The controlled ROS generation from the Ce6-HA hydrogels activated broader molecular pathways necessary for effective skin tissue repair. Therefore, ROS-triggering HA hydrogels could be a viable approach to accelerate recovery and reduce scarring in clinical settings.
Keywords: Angiogenesis; Hyaluronic acid; Hydrogel; Proliferation; Reactive oxygen species; Wound healing.
© 2025 The Authors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Synthesis of a retro-GFOGER Adamantane-Based Collagen Mimetic Peptide Imbibed in a Hyaluronic Acid Hydrogel for Enhanced Wound Healing.ACS Appl Bio Mater. 2025 Jun 16;8(6):4657-4672. doi: 10.1021/acsabm.4c01895. Epub 2025 Feb 19. ACS Appl Bio Mater. 2025. PMID: 39970309 Free PMC article.
-
A hydrogel crosslinked with mixed-valence copper nanoclusters for diabetic wound healing.Acta Biomater. 2025 Jun 15;200:340-357. doi: 10.1016/j.actbio.2025.05.044. Epub 2025 May 21. Acta Biomater. 2025. PMID: 40409507
-
Multifunctional protein-polysaccharide hydrogel with antimicrobial and angiogenic properties to promote infected wound healing.Biomater Adv. 2025 Dec;177:214387. doi: 10.1016/j.bioadv.2025.214387. Epub 2025 Jun 13. Biomater Adv. 2025. PMID: 40527064
-
Hydrogel dressings for venous leg ulcers.Cochrane Database Syst Rev. 2022 Aug 5;8(8):CD010738. doi: 10.1002/14651858.CD010738.pub2. Cochrane Database Syst Rev. 2022. PMID: 35930364 Free PMC article.
-
Dressings and topical agents for the management of open wounds after surgical treatment for sacrococcygeal pilonidal sinus.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013439. doi: 10.1002/14651858.CD013439.pub2. Cochrane Database Syst Rev. 2022. PMID: 35593897 Free PMC article.
References
-
- Auger F., Lacroix D., Germain L. Skin substitutes and wound healing. Skin Pharmacol. Physiol. 2009;22(2):94–102. - PubMed
-
- Berardesca E., Maibach H. Ethnic skin: overview of structure and function. J. Am. Acad. Dermatol. 2003;48(6):S139–S142. - PubMed
-
- Bhardwaj N., Chouhan D., B Mandal B. Tissue engineered skin and wound healing: current strategies and future directions. Curr. Pharm. Des. 2017;23(24):3455–3482. - PubMed
-
- Enoch S., Leaper D.J. Basic science of wound healing. Surgery (Oxf.) 2005;23(2):37–42.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

