Animal-free edible scaffolds from soy protein isolate for the scalable production of cultured meat
- PMID: 40689295
- PMCID: PMC12271769
- DOI: 10.1016/j.crfs.2025.101129
Animal-free edible scaffolds from soy protein isolate for the scalable production of cultured meat
Abstract
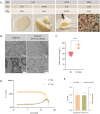
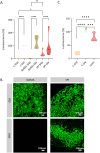
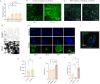
Large-scale production of cultured meat requires muscle cell culture in bioreactors, where microcarriers (MCs) support cell attachment, growth, and differentiation. However, most MCs are composed of inedible materials, requiring a cell detachment step, and/or contain animal-derived components, which are undesirable for cultured meat production. Therefore, we developed animal-free edible microcarriers based on soy protein isolate (SPI) that support muscle cell growth. SPI MCs supported cell attachment and growth similar to commercial collagen-coated dextran MCs, as bovine myoblasts expanded 24-fold over 8 days in a bioreactor. Moreover, myoblasts could differentiate into myotubes on the SPI-MCs. Importantly, SPI supported cell attachment in serum-free medium, as opposed to methacrylated gelatin (GelMA). Proteomics analysis revealed that, during SPI processing, cell adhesion peptides become available on the biomaterial, which also partially leach into the cell culture medium and replace serum components. To conclude, our study demonstrates the feasibility of growing and differentiating bovine muscle cells on edible, fully plant-based MCs, providing a scalable system for the production of cultured meat.
Keywords: Bovine myoblasts; Cultured meat; Microcarriers; Plant-based film; Proteomics; Soy protein isolate.
© 2025 The Authors. Published by Elsevier B.V.
Conflict of interest statement
None.
Figures


Similar articles
-
Development of Polysaccharide Hydrogels Enriched With Protein and Starch for Bovine Myoblast Cell Culture.J Food Sci. 2025 Jun;90(6):e70350. doi: 10.1111/1750-3841.70350. J Food Sci. 2025. PMID: 40556062
-
Immunomodulatory potential of dietary soybean-derived saponins.J Anim Sci. 2024 Jan 3;102:skae349. doi: 10.1093/jas/skae349. J Anim Sci. 2024. PMID: 39529449 Review.
-
A review on the effect of soy bioactive components on growth and health outcomes in pigs and broiler chickens.J Anim Sci. 2024 Jan 3;102:skae261. doi: 10.1093/jas/skae261. J Anim Sci. 2024. PMID: 39234891 Review.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
References
-
- Allan S.J., De Bank P.A., Ellis M.J. Bioprocess design considerations for cultured meat production with a focus on the expansion bioreactor. Front. Sustain. Food Syst. 2019;3 doi: 10.3389/fsufs.2019.00044. - DOI
LinkOut - more resources
Full Text Sources

