AKT signaling upregulates BDNF expression in induced neural stem cells that interact with microglia
- PMID: 40691843
- PMCID: PMC12282015
- DOI: 10.1186/s13287-025-04489-x
AKT signaling upregulates BDNF expression in induced neural stem cells that interact with microglia
Abstract
Background: Brain-derived neurotrophic factor (BDNF) has the capacity to promote neuronal survival that is crucial to neurological recovery after closed head injury (CHI). We previously reported that intracerebral-transplanted induced neural stem cells (iNSCs) can up-regulate BDNF levels to exert neurotrophic effects in CHI-damaged brains. Here we aim to elucidate the mechanism of BDNF up-regulation in iNSCs.
Methods: We performed iNSC and lipopolysaccharide (LPS)-activated microglia co-culture experiments, iNSC transplantation, loss-of-function study, morphological and molecular biological analyses to uncover the mechanism underlying the overexpression of BDNF in iNSCs.
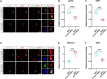
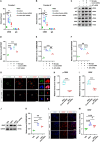
Results: Our results indicated that co-culture with LPS-activated microglia up-regulated the expression levels of BDNF, as well as Bdnf exons I and IV in iNSCs. Notably, AKT inhibition could counteract the effects of co-culture with LPS-activated microglia that decreased enhancer of zeste homolog 2 (EZH2) and H3K27 trimethylation (H3K27me3) levels at Bdnf promoter IV but increased EZH2 phosphorylation and BDNF expression in iNSCs. Additionally, blockage of AKT could counteract the effects of co-culture with LPS-activated microglia that increased cAMP response element binding protein (CREB) levels at Bdnf promoters I and IV, as well as CREB phosphorylation and BDNF expression in iNSCs. Furthermore, blocking AKT activity in grafted iNSCs could reduce BDNF expression in the injured cortices of CHI mice.
Conclusions: In short, our study shows that AKT signaling may regulate BDNF expression in iNSCs. Activation of AKT can up-regulate BDNF expression through inactivating EZH2 as well as reducing EZH2 and H3K27me3 levels at Bdnf promoter IV, meanwhile activating CREB as well as increasing CREB levels at Bdnf promoters I and IV.
Keywords: AKT; BDNF; CREB; Closed head injury; EZH2; H3K27me3; Induced neural stem cell; Microglia.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: (1) Title of the approved project: Effect and mechanism of induced neural stem cell transplantation in the treatment of brain injury; (2) Name of the institutional approval committee: Research Ethics Committee at Chinese PLA General Hospital; (3) The approval number: 2016-40; (4) The approval date: March 16, 2016. Consent for publication: Not applicable. Competing interests: All authors declare that they have no conflicts of interest.
Figures




Similar articles
-
Paeoniflorin exercise-mimetic potential regulates the Nrf2/HO-1/BDNF/CREB and APP/BACE-1/NF-κB/MAPK signaling pathways to reduce cognitive impairments and neuroinflammation in amnesic mouse model.Biomed Pharmacother. 2025 Aug;189:118299. doi: 10.1016/j.biopha.2025.118299. Epub 2025 Jun 30. Biomed Pharmacother. 2025. PMID: 40592207
-
[Acupuncture improves motor control by up-regulating expressions of BDNF and synaptic proteins in the striatum of neonatal rats with hypoxic-ischemic brain injury].Zhen Ci Yan Jiu. 2025 Jul 25;50(7):763-772. doi: 10.13702/j.1000-0607.20240690. Zhen Ci Yan Jiu. 2025. PMID: 40691026 Chinese.
-
MiR-134-5p/BDNF/TrkB/CREB signaling pathway involved in the depression-like behaviors in mice following exposure to benzo[a]pyrene.Metab Brain Dis. 2025 May 23;40(5):210. doi: 10.1007/s11011-025-01637-3. Metab Brain Dis. 2025. PMID: 40407953
-
Neuroplasticity - exercise-induced response of peripheral brain-derived neurotrophic factor: a systematic review of experimental studies in human subjects.Sports Med. 2010 Sep 1;40(9):765-801. doi: 10.2165/11534530-000000000-00000. Sports Med. 2010. PMID: 20726622
-
EORTC guidelines for the use of erythropoietic proteins in anaemic patients with cancer: 2006 update.Eur J Cancer. 2007 Jan;43(2):258-70. doi: 10.1016/j.ejca.2006.10.014. Epub 2006 Dec 19. Eur J Cancer. 2007. PMID: 17182241
References
-
- Maas AIR, Menon DK, Manley GT, Abrams M, Åkerlund C, Andelic N, Aries M, Bashford T, Bell MJ, Bodien YG, Brett BL, Büki A, Chesnut RM, Citerio G, Clark D, Clasby B, Cooper DJ, Czeiter E, Czosnyka M, Dams, Connor K, et al. Traumatic brain injury: progress and challenges in prevention, clinical care, and research. Lancet Neurol. 2022;21(11):1004–60. - PMC - PubMed
-
- Bielefeld P, Martirosyan A, Martín-Suárez S, Apresyan A, Meerhoff GF, Pestana F, Poovathingal S, Reijner N, Koning W, Clement RA, Van der Veen I, Toledo EM, Polzer O, Durá I, Hovhannisyan S, Nilges BS, Bogdoll A, Kashikar ND, Lucassen PJ, Belgard TG, et al. Traumatic brain injury promotes neurogenesis at the cost of astrogliogenesis in the adult hippocampus of male mice. Nat Commun. 2024;15(1):5222. - PMC - PubMed
-
- Lindhout FW, Krienen FM, Pollard KS, Lancaster MA. A molecular and cellular perspective on human brain evolution and tempo. Nature. 2024;630(8017):596–608. - PubMed
-
- Chaker Z, Segalada C, Kretz JA, Acar IE, Delgado AC, Crotet V, Moor AE, Doetsch F. Pregnancy-responsive pools of adult neural stem cells for transient neurogenesis in mothers. Science. 2023;382(6673):958–63. - PubMed
-
- Wyart C, Carbo-Tano M, Cantaut-Belarif Y, Orts-Del’Immagine A, Böhm UL. Cerebrospinal fluid-contacting neurons: multimodal cells with diverse roles in the CNS. Nat Rev Neurosci. 2023;24(9):540–56. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

