Diagnostic accuracy of ultrasound and MRI in parotid gland tumors: A retrospective study
- PMID: 40692766
- PMCID: PMC12277947
- DOI: 10.3892/etm.2025.12920
Diagnostic accuracy of ultrasound and MRI in parotid gland tumors: A retrospective study
Abstract
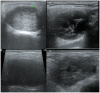
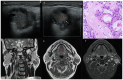
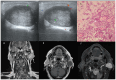
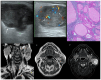
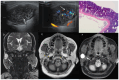
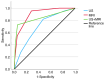
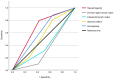
Parotid gland tumors (PGTs) are the most common salivary gland neoplasms, encompassing diverse benign and malignant pathologies. Accurate preoperative diagnosis is vital for surgical planning, functional outcomes (e.g., facial nerve preservation), and prognosis. While histopathology remains the gold standard, non-invasive imaging, such as ultrasound (US) and MRI, plays a critical role in initial tumor characterization. US is widely used due to its accessibility and cost-effectiveness, but its utility is constrained by acoustic limitations and operator dependence. MRI, with superior soft tissue contrast and multiplanar capabilities, excels in delineating tumor extent and neural involvement but often struggles to differentiate benign from malignant lesions due to overlapping imaging features. The comparisons of diagnostic performance between US and MRI limited, and optimal imaging parameters for specific PGT subtypes remain underinvestigated. The aim of the present study was to review and summarize the clinical presentations and histological types of parotid tumors, whilst also evaluating the diagnostic accuracy of US and MRI, to determine optimal US imaging parameters for pleomorphic adenomas and Warthin tumors. The medical records, including imaging examination results, type of parotidectomy and postoperative pathological findings, of 214 patients with confirmed PGTs were collected from the database of Central Hospital of Wuhan (Wuhan, China). The nature of the tumor was assessed based on imaging findings (US and MRI), with postoperative pathology (hematoxylin and eosin and immunohistochemistry) used as the gold standard. The sensitivity, specificity, accuracy, positive predictive value (PPV) and negative predictive value (NPV) for US and MRI were established. The area under the curve (AUC) was computed to compare the accuracy of US and MRI in identifying the nature of parotid gland tumors. The Youden index was calculated to evaluate the diagnostic power. Data analysis was conducted using SPSS version 29.0, where statistical significance was set at P<0.05 using the χ2 test. In the field of PGT diagnosis, the combination of US and MRI technologies was found to significantly enhance diagnostic precision. This integrated approach showed a statistically significant improvement over the use of US alone (P<0.05). Although MRI, as an independent modality, showed higher accuracy compared with that of US, the difference between MRI and US was not statistically significant. The receiver operating characteristic curve analysis indicated that the AUC for MRI in diagnosing benign and malignant parotid gland tumors was 0.899, which was significantly greater compared with that of US (0.702; P<0.001). The combined diagnosis using US and MRI achieved the highest specificity (94.8%), compared with US (52.6%) and MRI (71.9%) alone. For the combined US-MRI approach, the PPV, NPV and Youden index were 84.6, 95.0 and 0.68%. For US and MRI alone, the PPV, NPV and Youden index were 64.7, 94.1 and 0.34%, and 75.8, 95.0 and 0.67%, respectively. To conclude, these data suggest that the amalgamation of MRI and US can provide an efficacious means of diagnosing PGTs, thereby constituting an instrumentation for the preoperative qualitative evaluation of these tumors.
Keywords: MRI; parotid gland tumor; parotidectomy; ultrasound.
Copyright: © 2025 Kuang et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
123I-MIBG scintigraphy and 18F-FDG-PET imaging for diagnosing neuroblastoma.Cochrane Database Syst Rev. 2015 Sep 29;2015(9):CD009263. doi: 10.1002/14651858.CD009263.pub2. Cochrane Database Syst Rev. 2015. PMID: 26417712 Free PMC article.
-
Contrast-enhanced ultrasound using SonoVue® (sulphur hexafluoride microbubbles) compared with contrast-enhanced computed tomography and contrast-enhanced magnetic resonance imaging for the characterisation of focal liver lesions and detection of liver metastases: a systematic review and cost-effectiveness analysis.Health Technol Assess. 2013 Apr;17(16):1-243. doi: 10.3310/hta17160. Health Technol Assess. 2013. PMID: 23611316 Free PMC article.
-
Diagnostic test accuracy and cost-effectiveness of tests for codeletion of chromosomal arms 1p and 19q in people with glioma.Cochrane Database Syst Rev. 2022 Mar 2;3(3):CD013387. doi: 10.1002/14651858.CD013387.pub2. Cochrane Database Syst Rev. 2022. PMID: 35233774 Free PMC article.
-
Computed tomography versus magnetic resonance imaging versus bone scintigraphy for clinically suspected scaphoid fractures in patients with negative plain radiographs.Cochrane Database Syst Rev. 2015 Jun 5;2015(6):CD010023. doi: 10.1002/14651858.CD010023.pub2. Cochrane Database Syst Rev. 2015. PMID: 26045406 Free PMC article.
References
-
- Bonavolontà P, Dell'Aversana Orabona G, Maglitto F, Abbate V, Committeri U, Salzano G, Improta G, Iaconetta G, Califano L. Postoperative complications after removal of pleomorphic adenoma from the parotid gland: A long-term follow up of 297 patients from 2002 to 2016 and a review of publications. Br J Oral Maxillofac Surg. 2019;57:998–1002. doi: 10.1016/j.bjoms.2019.08.008. - DOI - PubMed
-
- Ferreira PC, Amarante JM, Rodrigues JM, Pinho CJ, Cardoso MA, Reis JC. Parotid surgery: Review of 107 tumors (1990-2002) Int Surg. 2005;90:160–166. - PubMed
LinkOut - more resources
Full Text Sources
