Immunomodulatory mechanisms of the gut microbiota and metabolites on regulatory T cells in rheumatoid arthritis
- PMID: 40692793
- PMCID: PMC12277365
- DOI: 10.3389/fimmu.2025.1610254
Immunomodulatory mechanisms of the gut microbiota and metabolites on regulatory T cells in rheumatoid arthritis
Abstract
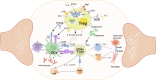
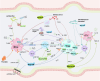
Rheumatoid arthritis (RA) is an autoimmune disease, in which the abnormal activation and proliferation of effector T cells play a pivotal role in its pathogenesis. Regulatory T cells (Tregs) are a unique subset of immune cells with immunosuppressive functions, which help to inhibit the differentiation and proliferation of effector T cells in RA and maintain immune tolerance. The interaction between gut microbiota and immune cells has long been a research hotspot in autoimmune diseases. Although gut microbiota metabolites are considered to regulate the host's immune system as a bridge of the gut-joint axis, how gut microbiota acts on immunosuppressive Tregs remains unclear. This review summarizes that how the gut microbiota directly or indirectly (via metabolites) enhances the immunosuppressive capacity of Tregs. This enhancement is primarily achieved through pathways such as promoting the induction of Tregs, upregulating the expression of characteristic transcription factors of Tregs, and facilitating their secretion of anti-inflammatory cytokines, thereby ameliorating the inflammatory microenvironment and subsequently improving autoimmune conditions in RA.
Keywords: autoimmune; gut microbiota; metabolites; regulatory T cells; rheumatoid arthritis.
Copyright © 2025 Xu, Zhou, Xie, Zhang, Gu, Liu, Liu and Chang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Microbiota-dependent modulation of intestinal anti-inflammatory CD4+ T cell responses.Semin Immunopathol. 2025 Apr 1;47(1):23. doi: 10.1007/s00281-025-01049-6. Semin Immunopathol. 2025. PMID: 40167791 Review.
-
Role of the intestinal flora-immunity axis in the pathogenesis of rheumatoid arthritis-mechanisms regulating short-chain fatty acids and Th17/Treg homeostasis.Mol Biol Rep. 2025 Jun 21;52(1):617. doi: 10.1007/s11033-025-10714-w. Mol Biol Rep. 2025. PMID: 40544212 Review.
-
Immunomodulatory properties of the gut microbiome: diagnostic and therapeutic potential for rheumatoid arthritis.Clin Exp Med. 2025 Jul 1;25(1):226. doi: 10.1007/s10238-025-01777-x. Clin Exp Med. 2025. PMID: 40591032 Free PMC article. Review.
-
Integrative multi-omics analysis reveals the interaction mechanisms between gut microbiota metabolites and ferroptosis in rheumatoid arthritis.Front Immunol. 2025 Jul 9;16:1608262. doi: 10.3389/fimmu.2025.1608262. eCollection 2025. Front Immunol. 2025. PMID: 40703513 Free PMC article.
-
Microbial imbalance in the gut: a new frontier in Rheumatoid arthritis research.Inflammopharmacology. 2025 May;33(5):2277-2291. doi: 10.1007/s10787-025-01737-7. Epub 2025 Apr 12. Inflammopharmacology. 2025. PMID: 40220199 Review.
References
-
- Nii T, Maeda Y, Motooka D, Naito M, Matsumoto Y, Ogawa T, et al. Genomic repertoires linked with pathogenic potency of arthritogenic Prevotella copri isolated from the gut of patients with rheumatoid arthritis. Ann Rheum Dis. (2023) 82:621–29. doi: 10.1136/ard-2022-222881, PMID: - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

