Microwave-enhanced additive-free C-H amination of benzoxazoles catalysed by supported copper
- PMID: 40692986
- PMCID: PMC12278112
- DOI: 10.3762/bjoc.21.108
Microwave-enhanced additive-free C-H amination of benzoxazoles catalysed by supported copper
Abstract
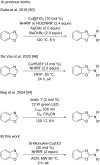
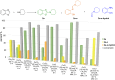
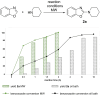
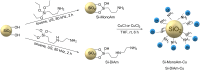
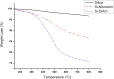
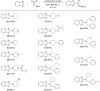
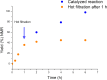
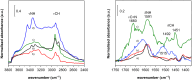
The C2-amination of benzoxazole offers wide-ranging potential for substrate expansion and the functionalisation of bioactive compounds. This study presents a green and efficient C-H amination, catalysed by CuCl and CuCl2, in acetonitrile without acidic, basic or oxidant additives that is accelerated by microwave (MW) irradiation and is completed in 1.5-2 h. A solid Cu(I) catalyst supported on aminated silica made the process cost-effective and heterogeneous, thus simplifying work-up and minimising free copper in solution. The catalyst was found to be regeneratable and reusable for up to eight cycles. The optimised method facilitated the synthesis of various benzoxazole derivatives, demonstrating its versatility and practical applicability.
Keywords: aerobic oxidation; copper; grafted silica; heterogeneous catalysis; microwave.
Copyright © 2025, Paraschiv et al.
Figures



References
-
- Abdullahi A, Yeong K Y. Med Chem Res. 2024;33:406–438. doi: 10.1007/s00044-024-03190-7. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
