Characteristics and availability of medicine early access programs and donations in Slovenia
- PMID: 40694381
- PMCID: PMC12282359
- DOI: 10.1093/oncolo/oyaf092
Characteristics and availability of medicine early access programs and donations in Slovenia
Abstract
Background: Early access programs (EAPs) and medicine donation programs (MDPs) enable patients to have access to new medicines prior to regulatory approval or national reimbursement, respectively. The objective of this study was to evaluate the characteristics and timeliness of EAPs and MDPs in Slovenia.
Methods: Originator medicines approved by the EMA through a centralized procedure from 2010 to 2019 were included in the study. Data on the EAPs and MDPs were obtained from the Agency for Medicinal Products and Medical Devices of Slovenia.
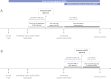
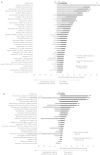
Results: The EMA approved 458 new indications for 324 medicines during the study period. In Slovenia, a total of 58 medicine indications (12.7%) became available before national reimbursement, including 35 (7.6%) by EAPs and 26 (5.7%) by MDPs (3 through both programs). Among these, 35 (60.3%) medicine indications were associated with oncology. EAPs facilitated access to medicines a median of 21.0 months before reimbursement (24.7 months for oncology medicines). Initiating EAPs at the time of the marketing authorization application could potentially improve the time of early access by an additional 5.5 months (5.2 months for oncology). MDPs enabled access to medicines 9.5 months prior to reimbursement (9.4 months for oncology), with potential further improvement by 7.9 months with the initiation of access at the time of marketing authorization (5.7 months for oncology).
Conclusion: Our findings highlight Slovenia's successful implementation of EAPs and MDPs, with oncology emerging as the predominant focal area. The potential exists for improvements in the scope, timeliness, and transparency of information on EAPs and MDPs accessible to patients in Slovenia.
Keywords: access time; antineoplastic and immunomodulating agents; compassionate use programs; early access programs; oncology.
© The Author(s) 2025. Published by Oxford University Press.
Conflict of interest statement
All authors declare potential conflicts of interest related to the research projects with European Medicines Agencies and the following companies: AstraZeneca, Krka, Novartis, and Swixx. Lea Knez has received a speaker honorarium from MSD, Pfizer, and Roche.
Figures
Similar articles
-
[Volume and health outcomes: evidence from systematic reviews and from evaluation of Italian hospital data].Epidemiol Prev. 2013 Mar-Jun;37(2-3 Suppl 2):1-100. Epidemiol Prev. 2013. PMID: 23851286 Italian.
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Assessment of the official national insurance coverage of regenerative medical products for ophthalmic diseases in Japan following regulatory approval.Regen Ther. 2025 Jul 12;30:384-388. doi: 10.1016/j.reth.2025.07.003. eCollection 2025 Dec. Regen Ther. 2025. PMID: 40689376 Free PMC article.
-
Interventions to improve safe and effective medicines use by consumers: an overview of systematic reviews.Cochrane Database Syst Rev. 2014 Apr 29;2014(4):CD007768. doi: 10.1002/14651858.CD007768.pub3. Cochrane Database Syst Rev. 2014. PMID: 24777444 Free PMC article.
References
-
- EC regulation Article 83. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32004R0726...
-
- Janzic U, Knez L, Janzic A, Cufer T.. Time to access to novel anticancer drugs and the correlation with ESMO-Magnitude of Clinical Benefit Scale in Slovenia. Expert Rev Pharmacoecon Outcomes Res. 2019;19:717-723. https://doi.org/ 10.1080/14737167.2019.1702879 - DOI - PubMed
-
- Jacquet E, Kerouani-Lafaye G, Grude F, et al. Comparative study on anticancer drug access times between FDA, EMA and the French temporary authorisation for use program over 13 years. Eur J Cancer. 2021;149:82-90. https://doi.org/ 10.1016/j.ejca.2021.03.008 - DOI - PubMed
-
- Balasubramanian G, Morampudi S, Chhabra P, Gowda A, Zomorodi B.. An overview of compassionate use programs in the european union member states. Vol. 5. Intractable and Rare Diseases Res. 2016;5:244-254. https://doi.org/ 10.5582/irdr.2016.01054 - DOI - PMC - PubMed
-
- Krendyukov A. Early Access Provision for Innovative Medicinal Products in Oncology: Challenges and Opportunities. Front Oncol. 2020;10:1604. https://doi.org/ 10.3389/fonc.2020.01604 - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

