Long-term effects of 4 years of menopausal hormone therapy on white matter integrity
- PMID: 40694740
- PMCID: PMC12382724
- DOI: 10.1097/GME.0000000000002562
Long-term effects of 4 years of menopausal hormone therapy on white matter integrity
Abstract
Objectives: To assess the long-term effects of 4 years of menopausal hormone therapy (mHT) on the brain's white matter architecture in women who initiated mHT within 3 years of menopause onset.
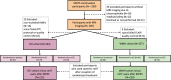
Methods: The Kronos Early Estrogen Prevention Study (KEEPS) was a multicenter, double-blind, randomized, placebo-controlled 4-year mHT trial with treatment arms of oral conjugated equine estrogens (oCEE), transdermal 17β-estradiol (tE2), and placebo in recently postmenopausal women. KEEPS Continuation was an observational follow-up of KEEPS participants. White matter integrity was evaluated in KEEPS Continuation participants 10 years after KEEPS completion using white matter hyperintensity volume, diffusion magnetic resonance imaging (dMRI) techniques, and cerebral infarcts. Linear regression models were fitted for each brain region to evaluate if there were differences in white matter between KEEPS treatment arms.
Results: There was no evidence to suggest the long-term effects of 4 years of mHT on brain white matter in KEEPS Continuation participants [n=266, mean age 67 (58-73)]. No differences in dMRI metrics were found in each of the treatment arms (oCEE n=70; tE2 n=79) when compared to placebo (n=94), following a false discovery rate adjustment for multiple comparisons. There were no statistically significant differences in white matter hyperintensity volume or infarct occurrence when comparing each of the treatment arms to placebo.
Conclusions: We found no evidence of the long-term effect of 4-year mHT on white matter integrity when compared to placebo, consistent with emerging evidence of the safety of short-term use of mHT in recently postmenopausal women.
Keywords: DTI; Diffusion MRI; Hormone therapy; Infarcts; NODDI; White matter hyperintensities..
Copyright © 2025 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of The Menopause Society.
Conflict of interest statement
Financial disclosure/conflicts of interest: K.K. served on the data safety monitoring board for Pfizer Inc. and Takeda Global Research & Development Center, Inc. She received research support from Avid Radiopharmaceuticals and Eli Lilly. She consults for Biogen. N.S. is a member of the Scientific Advisory Boards for Astellas, Que Oncology, Amazon (Ember), and Menogenix, Inc. She is a consultant for Ansh Labs, and has received grant support (to her institution) from Menogenix, Inc. She has been paid by Fertility IQ for developing a series of videos on menopause and has stock options (no payments) from Menogenix, Inc. L.P. is a member of the advisory board for Flo Health and has a financial relationship with WinFertility. E.K. is funded in part by the National Institute on Aging (NIA grant U54 AG044170). E.K. has no conflicts of interest directly related to the subject of this manuscript. However, over the past 36 months, she has had the following conflicts of interest: she has been a consultant for Astellas and Mithra Pharmaceuticals, Scynexis, and Womaness. She receives grant support from Mithra Pharmaceuticals. She has received payment for the development of educational content from Med Learning Group and the Academy of Continued Healthcare Learning. She has received honoraria for CME activity from PriMed and OBG Management. A.J.F. received funding from Northwestern University for participating in a DSMB ($500 in the last year). C.G.S. received past funding from Karolinska Institute and Drum Tower Hospital, Nanjing. J.A.F. receives ongoing institutional funding from the National Institute of Health and Mangurian Foundation and receives ongoing personal funding from the University of Pennsylvania and Medtronic Inc. T.T.J. was one of 2 principal investigators on a grant from the Alzheimer’s Clinical Trial Consortium (ACTC). D.B.H. received institutional funding from the NIH and Alzheimer’s Association funding. M.M.-A. received past funding from the Biomedical Research Alliance of New York. The other authors have nothing to disclose.
Figures



References
-
- Karamitrou EK, Anagnostis P, Vaitsi K, Athanasiadis L, Goulis DG. Early menopause and premature ovarian insufficiency are associated with increased risk of dementia: a systematic review and meta-analysis of observational studies. Maturitas 2023;176:107792. doi: 10.1016/j.maturitas.2023.107792 - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

