Exogenous arginine differentially regulates inflammatory cytokine and inducible nitric oxide synthase expression in macrophages
- PMID: 40694828
- PMCID: PMC12282984
- DOI: 10.1093/immhor/vlaf028
Exogenous arginine differentially regulates inflammatory cytokine and inducible nitric oxide synthase expression in macrophages
Abstract
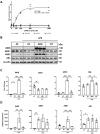
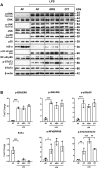
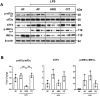
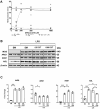
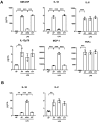
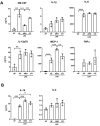
Immune dysfunction and late mortality from multiorgan failure are hallmarks of severe sepsis. Arginine, a semi-essential amino acid important for protein synthesis, immune response, and circulatory regulation, is deficient in sepsis. However, arginine supplementation in sepsis remains controversial due to the potential to upregulate inducible nitric oxide synthase (iNOS)-mediated excessive nitric oxide (NO) generation in macrophages, leading to vasodilation and hemodynamic catastrophe. Citrulline supplementation has been considered an alternative to replenishing arginine via de novo synthesis, orchestrated by argininosuccinate synthase 1 (ASS1) and argininosuccinate lyase (ASL). However, the functional relevance of the ASS1-ASL pathway in macrophages after endotoxin stimulation is unclear but it is crucial to consider amino acid restoration as a tool for treating sepsis. We demonstrate that lipopolysaccharide (LPS)-mediated iNOS, ASS1, and ASL protein expression and nitric oxide generation were dependent on exogenous arginine in RAW 264.7 macrophages. Exogenous citrulline was not sufficient to restore nitric oxide generation in arginine-free conditions. Despite the induction of iNOS and ASS1 mRNA in arginine-free conditions, exogenous arginine was necessary and citrulline was not sufficient to overcome eIF2-α (elongation initiation factor 2-α)-mediated translational repression of iNOS and ASS1 protein expression. Moreover, exogenous arginine, but not citrulline, selectively modified the inflammatory cytokine and chemokine expression profile of the LPS-activated RAW 264.7 and bone marrow-derived macrophages. Our study highlights the complex, differential regulation of proinflammatory cytokine expression, and NO generation by exogenous arginine in macrophages.
Keywords: cell activation; cytokines; endotoxin shock; monocytes/macrophages; nitric oxide.
© The Author(s) 2025. Published by Oxford University Press on behalf of The American Association of Immunologists.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Oncogenic Kaposi's Sarcoma-Associated Herpesvirus Upregulates Argininosuccinate Synthase 1, a Rate-Limiting Enzyme of the Citrulline-Nitric Oxide Cycle, To Activate the STAT3 Pathway and Promote Growth Transformation.J Virol. 2019 Feb 5;93(4):e01599-18. doi: 10.1128/JVI.01599-18. Print 2019 Feb 15. J Virol. 2019. PMID: 30463977 Free PMC article.
-
Lacticaseibacillus rhamnosus GG-driven remodeling of arginine metabolism mitigates gut barrier dysfunction.Am J Physiol Gastrointest Liver Physiol. 2025 Jul 1;329(1):G162-G185. doi: 10.1152/ajpgi.00366.2024. Epub 2025 May 26. Am J Physiol Gastrointest Liver Physiol. 2025. PMID: 40418622 Free PMC article.
-
Sustained generation of nitric oxide and control of mycobacterial infection requires argininosuccinate synthase 1.Cell Host Microbe. 2012 Sep 13;12(3):313-23. doi: 10.1016/j.chom.2012.07.012. Cell Host Microbe. 2012. PMID: 22980328 Free PMC article.
-
Citrullinemia Type I.2004 Jul 7 [updated 2022 Aug 18]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2004 Jul 7 [updated 2022 Aug 18]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 20301631 Free Books & Documents. Review.
-
Interventions affecting the nitric oxide pathway versus placebo or no therapy for fetal growth restriction in pregnancy.Cochrane Database Syst Rev. 2023 Jul 10;7(7):CD014498. doi: 10.1002/14651858.CD014498. Cochrane Database Syst Rev. 2023. PMID: 37428872 Free PMC article.
References
-
- Gotts JE, Matthay MA. Sepsis: pathophysiology and clinical management. BMJ. 2016;353:i1585. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

