Importance of landscape exploration and progress in molecular therapies and precision medicine for pancreatic ductal adenocarcinoma
- PMID: 40697241
- PMCID: PMC12278137
- DOI: 10.4251/wjgo.v17.i7.103337
Importance of landscape exploration and progress in molecular therapies and precision medicine for pancreatic ductal adenocarcinoma
Abstract
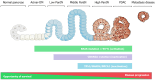
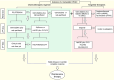
Pancreatic ductal adenocarcinoma (PDAC) is a global health challenge and remains one of the most lethal malignancies; there are only a few therapeutic options. However, significant efforts have led to the identification of major genetic factors that drive the progression and pathogenesis of PDAC. Notably, the research and application of molecular targeted therapies and immunotherapies have rapidly increased and facilitated great progress in the treatment of many malignant tumors, additional targeted therapies and immunotherapy based on next-generation sequencing results provide new opportunities for the diagnosis and treatment of pancreatic tumors. Immune checkpoint inhibitors are also now being incorporated as PDAC therapies, and combinations of molecularly targeted therapies with immunotherapies are emerging as strategies for boosting the immune response. The investigation of biomarkers of a response or primary resistance to immunotherapies is also an emerging research area. Herein, we further discuss the recent technological advances that continue to expand our understanding of PDAC complexity. We discuss the advancements expected in the near future, including biomarker-driven treatments and immunotherapies. We presume that the clinical translation of these research efforts will improve the survival outcomes of this challenging disease, which are currently dismal.
Keywords: Immunotherapy; Microenvironment; Molecular subtypes; Pancreatic ductal adenocarcinoma; Precision medicine; Therapeutic targets; Transcriptomic.
©The Author(s) 2025. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Figures



References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7–34. - PubMed
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33. - PubMed
-
- Conroy T, Hammel P, Hebbar M, Ben Abdelghani M, Wei AC, Raoul JL, Choné L, Francois E, Artru P, Biagi JJ, Lecomte T, Assenat E, Faroux R, Ychou M, Volet J, Sauvanet A, Breysacher G, Di Fiore F, Cripps C, Kavan P, Texereau P, Bouhier-Leporrier K, Khemissa-Akouz F, Legoux JL, Juzyna B, Gourgou S, O'Callaghan CJ, Jouffroy-Zeller C, Rat P, Malka D, Castan F, Bachet JB Canadian Cancer Trials Group and the Unicancer-GI–PRODIGE Group. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N Engl J Med. 2018;379:2395–2406. - PubMed
-
- Gobbi PG, Bergonzi M, Comelli M, Villano L, Pozzoli D, Vanoli A, Dionigi P. The prognostic role of time to diagnosis and presenting symptoms in patients with pancreatic cancer. Cancer Epidemiol. 2013;37:186–190. - PubMed
-
- Sohal DPS, Kennedy EB, Khorana A, Copur MS, Crane CH, Garrido-Laguna I, Krishnamurthi S, Moravek C, O'Reilly EM, Philip PA, Ramanathan RK, Ruggiero JT, Shah MA, Urba S, Uronis HE, Lau MW, Laheru D. Metastatic Pancreatic Cancer: ASCO Clinical Practice Guideline Update. J Clin Oncol. 2018;36:2545–2556. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

