Safety and toxicity risks of radiotherapy combined with PD-1/PD-L1 inhibitors: A comprehensive review
- PMID: 40697412
- PMCID: PMC12281118
- DOI: 10.1016/j.isci.2025.112882
Safety and toxicity risks of radiotherapy combined with PD-1/PD-L1 inhibitors: A comprehensive review
Abstract
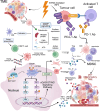
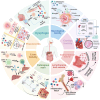
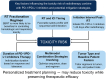
Radiotherapy (RT) combined with PD-1/PD-L1 Inhibitors has demonstrated remarkable efficacy across various cancers. However, concerns remain regarding its safety and potential toxicity. We analyze toxicity risks of RT combined with anti-PD-1/PD-L1 therapy based on current clinical studies. While a slight increase in grade 1-2 pneumonitis has been reported in patients with lung cancer, no consistent evidence suggests a rise in ≥ grade 3 pulmonary toxicity. Additionally, the combination appears well tolerated in the central nervous system, head and neck, and hepatic malignancies. This narrative review investigates key factors that may influence the risk of toxicity in combination therapy, including the dose and fractionation of RT, sequencing with immunotherapy, timing and duration of immune consolidation, and regimen heterogeneity. This review adheres to the SANRA (Scale for the Assessment of Narrative Review Articles) guidelines.
Keywords: Immune response; Oncology; Therapy.
© 2025 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Interventions to reduce acute and late adverse gastrointestinal effects of pelvic radiotherapy for primary pelvic cancers.Cochrane Database Syst Rev. 2018 Jan 23;1(1):CD012529. doi: 10.1002/14651858.CD012529.pub2. Cochrane Database Syst Rev. 2018. PMID: 29360138 Free PMC article.
-
Efficacy and safety of first-line PD-1/PD-L1 inhibitors combined with or without anti-angiogenesis therapy for extensive-stage small-cell lung cancer: a network meta-analysis.Ther Adv Med Oncol. 2025 Jun 25;17:17588359251348310. doi: 10.1177/17588359251348310. eCollection 2025. Ther Adv Med Oncol. 2025. PMID: 40574965 Free PMC article.
-
Comparison of efficacy and safety of PD-1/PD-L1 combination therapy in first-line treatment of advanced NSCLC: an updated systematic review and network meta-analysis.Clin Transl Oncol. 2024 Oct;26(10):2488-2502. doi: 10.1007/s12094-024-03442-3. Epub 2024 Apr 16. Clin Transl Oncol. 2024. PMID: 38625495
-
Treatment of newly diagnosed glioblastoma in the elderly: a network meta-analysis.Cochrane Database Syst Rev. 2020 Mar 23;3(3):CD013261. doi: 10.1002/14651858.CD013261.pub2. Cochrane Database Syst Rev. 2020. PMID: 32202316 Free PMC article.
-
Impact of delayed addition of PD-1/PD-L1 inhibitors to chemotherapy on outcomes in patients with extensive-stage small cell lung cancer.Ther Adv Med Oncol. 2025 Jul 20;17:17588359251356919. doi: 10.1177/17588359251356919. eCollection 2025. Ther Adv Med Oncol. 2025. PMID: 40697877 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

