Pharmacological management of type 2 diabetes mellitus in children and adolescents: A systematic review and network meta-analysis
- PMID: 40697609
- PMCID: PMC12278083
- DOI: 10.4239/wjd.v16.i7.106890
Pharmacological management of type 2 diabetes mellitus in children and adolescents: A systematic review and network meta-analysis
Abstract
Background: The incidence of type 2 diabetes mellitus (T2DM) in children and adolescents is increasing, yet there is limited information on the available pharmacological interventions to combat T2DM and prevent associated comorbidities.
Aim: To assess the effectiveness of current pharmacological treatments in managing T2DM in children and adolescents. The protocol of the study was registered in PROSPERO (CRD42022382165).
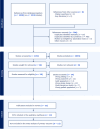
Methods: Searches were performed in PubMed, EMBASE, Scopus, and ClinicalTrials.gov for publications between 1990 to September 2024 without language restrictions. Randomized control trials (RCTs) of pharmacotherapy in children and adolescents with T2DM (aged < 19 years) were included. The primary outcome was a change in glycated hemoglobin (HbA1c) from baseline to follow-up. Secondary outcomes were changes in body weight, body mass index (BMI), total cholesterol, triglycerides, high density lipoprotein, and low-density lipoprotein from baseline, and incidence of adverse events during study periods. Screening, full-text review, data extraction, and assessments of risk of bias were done by two reviewers. Conflicts on each step were resolved by a third reviewer. Data analysis was performed using Review Manager Version 6.5 (RevMan 6.5) and 'R' software via RStudio, 'meta' and 'netmeta'.
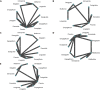
Results: A total of 12 studies having low to moderate risk of bias with 1658 participants, and follow-up duration 12-52 weeks were included. In our network meta-analysis, compared to control(s), the reduction of HbA1c was significantly larger for dulaglutide [mean difference (MD), 95% confidence interval: -1.20, -2.12 to -0.28], followed by dapagliflozin (-0.94, -1.44 to -0.44), liraglutide (-0.91, -1.37 to -0.45), empagliflozin (-0.87, -1.40 to -0.34), exenatide (-0.59, -1.07 to -0.11) and linagliptin (-0.45, -0.87 to -0.02) while other drugs had little or no effect. While liraglutide was associated with a change in body weight [MD -2.41 (-4.68, -0.14) kg], no other drug treatment was associated with significant changes in body weight, BMI, and lipids. Apart from level 1 hypoglycemia with liraglutide [risk difference (RD): 0.20, 0.04-0.37] and minor adverse events with dulaglutide (RD: 0.24, 0.08-0.40), no other treatment was associated with excess risk of hypoglycemia or minor or major adverse events.
Conclusion: Pharmacotherapy of T2DM with dulaglutide, dapagliflozin, liraglutide, empagliflozin, exenatide, and linagliptin in children is associated with modest reduction of HbA1c. Larger RCTs with longer follow-up durations are needed to guide better therapeutic decision making.
Keywords: Adolescents; Children; Diabesity; Glycemic control; Obesity; Pharmacotherapy; Type 2 diabetes mellitus.
©The Author(s) 2025. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Figures

References
-
- Wagenknecht LE, Lawrence JM, Isom S, Jensen ET, Dabelea D, Liese AD, Dolan LM, Shah AS, Bellatorre A, Sauder K, Marcovina S, Reynolds K, Pihoker C, Imperatore G, Divers J SEARCH for Diabetes in Youth study. Trends in incidence of youth-onset type 1 and type 2 diabetes in the USA, 2002-18: results from the population-based SEARCH for Diabetes in Youth study. Lancet Diabetes Endocrinol. 2023;11:242–250. - PMC - PubMed
-
- Hawton K, Apperley L, Parkinson J, Owens M, Semple C, Canvin L, Holt A, Easter S, Clark K, Lund K, Clarke E, O'Brien J, Giri D, Senniappan S, Shield JPH. Complications of excess weight seen in two tier 3 paediatric weight management services: an observational study. Arch Dis Child. 2025;110:216–220. - PubMed
-
- Hampl SE, Hassink SG, Skinner AC, Armstrong SC, Barlow SE, Bolling CF, Avila Edwards KC, Eneli I, Hamre R, Joseph MM, Lunsford D, Mendonca E, Michalsky MP, Mirza N, Ochoa ER, Sharifi M, Staiano AE, Weedn AE, Flinn SK, Lindros J, Okechukwu K. Executive Summary: Clinical Practice Guideline for the Evaluation and Treatment of Children and Adolescents With Obesity. Pediatrics. 2023;151:e2022060641. - PubMed
-
- Hannon TS, Arslanian SA. Obesity in Adolescents. N Engl J Med. 2023;389:251–261. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

