Sustained high expression of human FVII following AAV8-mediated gene delivery in mice
- PMID: 40697775
- PMCID: PMC12282199
- DOI: 10.1016/j.omtm.2025.101523
Sustained high expression of human FVII following AAV8-mediated gene delivery in mice
Abstract
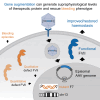
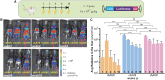
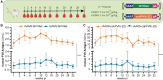
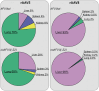
Factor VII (FVII) deficiency is a rare bleeding disorder with a prevalence of approximately 1:300,000-500,000 individuals. We explored whether adeno-associated virus (AAV)-mediated gene therapy can achieve durable and functional expression of human FVII (hFVII) in vivo. Wild-type hFVII (hFVIIwt) and a naturally occurring splice variant designated hFVII(-22) (GenBank: NM_019616.4) were expressed under the control of the Apolipoprotein E-derived hepatic locus control region and the α1-anti-trypsin promoter. Expression cassettes were packaged in either recombinant AAV5 (rAAV5) or recombinant AAV8 (rAAV8). For both hFVIIwt and hFVII(-22), 5- to 20-fold higher plasma levels of hFVII could be obtained when rAAV8 was used as a vector as opposed to rAAV5. Interestingly, hFVII levels obtained by employing rAAV8 expressing the hFVII(-22) cDNA variant were approximately 10 times higher than those obtained using rAAV8 expressing hFVIIwt. Based on these results, we generated an rAAV8-based gene therapy vector encoding hFVII(-22) and evaluated long-term expression in vivo. Employing a vector dose of 0.8 × 1012 genome copies (gc)/kg, we observed 48 weeks of functional hFVII expression which peaked at 16 IU/mL and stabilized at 7 IU/mL. These results support the pre-clinical development of AAV8-mediated delivery employing the splice-variant hFVII(-22) for patients suffering from FVII deficiency.
Keywords: AAV5; AAV8; FVII deficiency; augmentation therapy; gene therapy; liver-directed gene therapy; rare bleeding disorders.
© 2025 The Authors.
Conflict of interest statement
A.D., B.B., A.v.A., J. Verhaagen, D.G.M.M., and J. Voorberg are listed as inventors on a patent application on the expression of therapeutic genes, including FVII, for gene therapy applications.
Figures



Similar articles
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
Can a Liquid Biopsy Detect Circulating Tumor DNA With Low-passage Whole-genome Sequencing in Patients With a Sarcoma? A Pilot Evaluation.Clin Orthop Relat Res. 2025 Jan 1;483(1):39-48. doi: 10.1097/CORR.0000000000003161. Epub 2024 Jun 21. Clin Orthop Relat Res. 2025. PMID: 38905450
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Sertindole for schizophrenia.Cochrane Database Syst Rev. 2005 Jul 20;2005(3):CD001715. doi: 10.1002/14651858.CD001715.pub2. Cochrane Database Syst Rev. 2005. PMID: 16034864 Free PMC article.
-
Progesterone or progestogen-releasing intrauterine systems for heavy menstrual bleeding.Cochrane Database Syst Rev. 2015 Apr 30;(4):CD002126. doi: 10.1002/14651858.CD002126.pub3. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2020 Jun 12;6:CD002126. doi: 10.1002/14651858.CD002126.pub4. PMID: 25924648 Updated.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

