Applying Field and Genomic Epidemiology Methods to Investigate Transmission Networks of Highly Pathogenic Avian Influenza A (H5N1) in Domestic Poultry in British Columbia, Canada (2022-2023)
- PMID: 40697881
- PMCID: PMC12283179
- DOI: 10.1155/tbed/4099285
Applying Field and Genomic Epidemiology Methods to Investigate Transmission Networks of Highly Pathogenic Avian Influenza A (H5N1) in Domestic Poultry in British Columbia, Canada (2022-2023)
Abstract
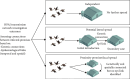
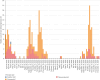
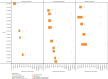
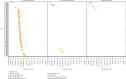
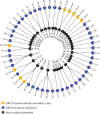
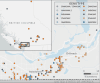
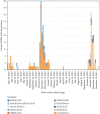
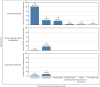
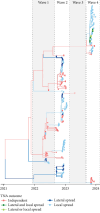
Highly pathogenic avian influenza (HPAI) virus A (H5N1) was first detected in North America in 2021. Ongoing spillover events from wild to domestic birds and subsequent transmission between domestic birds resulted in Canada's largest recorded epidemic of HPAI. Between December 2021 and April 2024, 422 A (H5N1) outbreaks in domestic birds were confirmed in Canada. Of these, 158 (37%) occurred in British Columbia (BC). This study integrates field and genomic epidemiology (GE) results to investigate the potential for lateral or local spread between infected poultry farms in BC. Five genetically distinct viral clusters were identified during this period. Among the 31 noncommercial premises, 27 (87.1%) were classified as independent introductions, while four premises (12.9%) were attributed to local spread within 10 km of a phylogenetically connected infected premises (IP). No lateral spread events were identified among noncommercial premises. All infected noncommercial premises housed birds with outdoor access, emphasizing their susceptibility to wild bird exposure. Of the 127 infected commercial poultry premises, 21 (16.5%) were classified as independent introductions, 82 (64.6%) as local spread, 18 (14.2%) with potential for lateral spread, five (3.9%) with potential for both local and/or lateral spread, and one (0.8%) for which sequencing was unavailable. Local spread emerged as a prominent feature, with most IP in proximity to one another having genetically similar viruses. Results suggest that proximity (<200 m) to an IP was a more reliable predictor of future infection status than contact with an IP. These findings underscore the critical value of combining field and GE to understand outbreak dynamics comprehensively. This integrative approach improves resource allocation, informs targeted containment strategies, and supports the need for effective biosecurity measures to mitigate future risks, particularly in densely populated poultry production regions. Robust interventions are needed to address both independent introductions and secondary spread pathways.
Copyright © 2025 Krista Howden et al. Transboundary and Emerging Diseases published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Comparative pathogenicity of three A(H5N1) clade 2.3.4.4b HPAI viruses in blue-winged teal and transmission to domestic poultry.mSphere. 2025 Jun 25;10(6):e0002125. doi: 10.1128/msphere.00021-25. Epub 2025 May 22. mSphere. 2025. PMID: 40401902 Free PMC article.
-
Vaccination and surveillance for high pathogenicity avian influenza in poultry-current situation and perspectives.Biologicals. 2025 Aug;91:101840. doi: 10.1016/j.biologicals.2025.101840. Epub 2025 May 13. Biologicals. 2025. PMID: 40359590
-
Dynamics of a Panzootic: Genomic Insights, Host Range, and Epidemiology of the Highly Pathogenic Avian Influenza A(H5N1) Clade 2.3.4.4b in the United States.Viruses. 2025 Feb 25;17(3):312. doi: 10.3390/v17030312. Viruses. 2025. PMID: 40143242 Free PMC article. Review.
-
Highly pathogenic avian influenza (H5N1): pathways of exposure at the animal-human interface, a systematic review.PLoS One. 2011 Jan 24;6(1):e14582. doi: 10.1371/journal.pone.0014582. PLoS One. 2011. PMID: 21283678 Free PMC article.
-
A Systematic Review of the Comparative Epidemiology of Avian and Human Influenza A H5N1 and H7N9 - Lessons and Unanswered Questions.Transbound Emerg Dis. 2016 Dec;63(6):602-620. doi: 10.1111/tbed.12327. Epub 2015 Jan 29. Transbound Emerg Dis. 2016. PMID: 25644240
References
-
- Andrew C. L., Russell S. L., Coombe M., et al. Descriptive Epidemiology and Phylodynamics of the “First Wave” of an Outbreak of Highly Pathogenic Avian Influenza (H5N1 Clade 2.3.4.4b) in British Columbia and the Yukon, Canada, April to September 2022. Transboundary and Emerging Diseases . 2024;2024(1) doi: 10.1155/2024/2327939.2327939 - DOI - PMC - PubMed
-
- Russell S. L., Andrew C. L., Yang K. C., et al. Descriptive Epidemiology and Phylogenetic Analysis of Highly Pathogenic Avian Influenza H5N1 Clade 2.3.4.4b in British Columbia (B.C.) and the Yukon, Canada, September 2022 to June 2023. Emerging Microbes & Infections . 2024;13(1) doi: 10.1080/22221751.2024.2392667. - DOI - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

