Molecular Assessment of Traditional Mongolian Medicine Tongola-5 in Lactulose Induced Diarrhea: An in vivo Study
- PMID: 40698060
- PMCID: PMC12282606
- DOI: 10.2147/DDDT.S527657
Molecular Assessment of Traditional Mongolian Medicine Tongola-5 in Lactulose Induced Diarrhea: An in vivo Study
Abstract
Propose: To elucidate the mechanisms underlying the therapeutic effects of Tonglaga-5 (TLG5) in the treatment of chronic diarrhea.
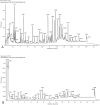
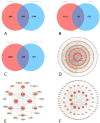
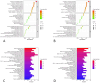
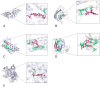
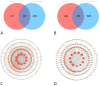
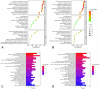
Methods: The chemical compositions of TLG5 in both in vitro and systemic circulation were analyzed using UPLC-Q/TOF MSE. Network pharmacology was applied to predict the therapeutic effects of TLG5 on chronic diarrhea. Molecular docking was utilized to assess the binding affinity between key bioactive compounds and their corresponding targets. A total of 24 male Sprague-Dawley (SD) rats were randomly assigned to three groups: control, model, and TLG5 treatment. Corresponding administration included a standard diet, a high-lactose diet of varying concentrations, and TLG5 by gavage during modeling. Transcriptomic analysis was employed to assess gene expression changes in intestinal tissues. Alterations in the intestinal microbiota were evaluated using 16S rRNA sequencing. qPCR and immunohistochemistry technologies were adopted to analyze the expression of genes and proteins associated with intestinal barrier integrity. ELISA was performed to quantify levels of bile acids and short-chain fatty acids (SCFAs).
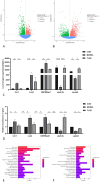
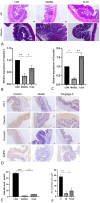
Results: UPLC-Q/TOF MSE identified 118 compounds in vitro and 52 in the systemic circulation. Network pharmacological analysis revealed that the in vitro and in vivo components of TLG5 were associated with five core targets and five key compounds, respectively. RNA sequencing analysis showed that TLG5 treatment inhibited the inflammatory pathway. 16S rRNA sequencing demonstrated that TLG5 administration led to an upregulation of beneficial microbial populations and a concomitant downregulation of pathogenic bacteria. The TLG5 treatment group presented a reduction in necrotic areas and inflammatory cell infiltration, as well as preservation of the mucosal structure, with a marked decline in inflammatory lesions. Moreover, TLG5 treatment resulted in a significant increase in the levels of SCFAs and bile acids in the cecum.
Conclusion: TLG5 exerts therapeutic effects on chronic diarrhea through multiple mechanisms, including modulation of intestinal microbiota diversity, enhancement of intestinal barrier function, and attenuation of inflammatory responses.
Keywords: Mongolian medicine Tonglaga-5; chronic diarrhea; intestinal barrier; intestinal microbiota.
© 2025 Ma et al.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures





Similar articles
-
Sishen pill alleviates ulcerative colitis via the NLRP3/ASC/Caspase-1 signaling pathway: Comprehensive validation through UPLC-Q-TOF/MS, network pharmacology, molecular docking, and in vivo experiments.J Chromatogr B Analyt Technol Biomed Life Sci. 2025 Oct 1;1264:124735. doi: 10.1016/j.jchromb.2025.124735. Epub 2025 Jul 13. J Chromatogr B Analyt Technol Biomed Life Sci. 2025. PMID: 40675106
-
Investigation of the effect and mechanism of Fei Re Pu Qing powder in treating acute lung injury (ALI) by modulating macrophage polarization via serum pharmacology and network pharmacology.J Ethnopharmacol. 2025 Jul 24;351:120089. doi: 10.1016/j.jep.2025.120089. Epub 2025 Jun 9. J Ethnopharmacol. 2025. PMID: 40499803
-
KunMingShanHaiTang formula reprograms macrophage metabolism and promotes M2 polarization via the HIF-1α pathway to alleviate ulcerative colitis symptoms in a rat model.J Bioenerg Biomembr. 2025 Jun;57(2-3):119-145. doi: 10.1007/s10863-025-10056-z. Epub 2025 Apr 2. J Bioenerg Biomembr. 2025. PMID: 40172736 Free PMC article.
-
Gut microbiome-based interventions for the management of obesity in children and adolescents aged up to 19 years.Cochrane Database Syst Rev. 2025 Jul 10;7(7):CD015875. doi: 10.1002/14651858.CD015875. Cochrane Database Syst Rev. 2025. PMID: 40637175 Review.
-
Probiotics for the prevention of pediatric antibiotic-associated diarrhea.Cochrane Database Syst Rev. 2011 Nov 9;(11):CD004827. doi: 10.1002/14651858.CD004827.pub3. Cochrane Database Syst Rev. 2011. Update in: Cochrane Database Syst Rev. 2015 Dec 22;(12):CD004827. doi: 10.1002/14651858.CD004827.pub4. PMID: 22071814 Updated.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

