Efficacy of irreversible electroporation combined with immunotherapy versus irreversible electroporation alone in locally advanced pancreatic cancer: a propensity score-matched retrospective study
- PMID: 40698084
- PMCID: PMC12279750
- DOI: 10.3389/fimmu.2025.1620988
Efficacy of irreversible electroporation combined with immunotherapy versus irreversible electroporation alone in locally advanced pancreatic cancer: a propensity score-matched retrospective study
Abstract
Background: Irreversible electroporation (IRE) has shown promise in improving survival outcomes and activating the immune response in patients with locally advanced pancreatic cancer (LAPC). Given these immune-enhancing effects, we hypothesized that combining IRE with immune checkpoint inhibitors may further improve treatment outcomes. This study aimed to evaluate the efficacy and safety of IRE combined with anti-PD-1 immunotherapy versus IRE alone in patients with LAPC.
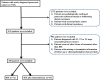
Methods: In this retrospective study, LAPC patients treated either with IRE plus toripalimab (240 mg administered 7 days post-IRE) or with IRE alone were included. Propensity score matching (PSM) analyses were employed for analysis. Clinical outcomes including overall survival (OS), progression-free survival (PFS), and treatment-related adverse events were analyzed and compared between the groups.
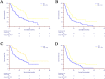
Results: A total of 108 patients from August 2015 and Match 2024 from SYSUCC cohort were identified with 76 undergoing IRE and 32 undergoing IRE and toripalimab in this study. After PSM, 96 patients consisting of 64 and 32 patients in the IRE and combination groups were enrolled. Clinical factors were all balanced between two groups. Patients receiving IRE combined with toripalimab showed significantly improved OS (35.03 months; 95% CI: 30.94-39.13 vs. 15.87 months; 95% CI: 8.99-22.74; P=0.014) and PFS (14.33months; 95% CI: 11.19-17.47 vs. 7.47 months; 95% CI: 3.86-11.08; P=0.022) compared to those receiving IRE alone. No treatment-related mortality was reported in either group and no statistically significant differences were observed in terms of complications and adverse events between two groups (all P>0.05).
Conclusions: The combination of IRE and anti-PD-1 immunotherapy was associated with improved survival outcomes and acceptable safety profiles compared to IRE alone in patients with LAPC. Further investigation through prospective trials is warranted.
Keywords: efficacy; immunotherapy; irreversible electroporation; locally advanced pancreatic cancer; prognosis.
Copyright © 2025 Xi, Sun, Chen, Yao, Zhu, Li and He.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Mukherjee S, Hurt CN, Bridgewater J, Falk S, Cummins S, Wasan H, et al. Radhakrishna G et al: Gemcitabine-based or capecitabine-based chemoradiotherapy for locally advanced pancreatic cancer (SCALOP): a multicentre, randomised, phase 2 trial. Lancet Oncol. (2013) 14:317–26. doi: 10.1016/S1470-2045(13)70021-4, PMID: - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

