Complement and coagulation cascade cross-talk in endometriosis and the potential of Janus Kinase inhibitors-a network meta-analysis
- PMID: 40698088
- PMCID: PMC12279843
- DOI: 10.3389/fimmu.2025.1619434
Complement and coagulation cascade cross-talk in endometriosis and the potential of Janus Kinase inhibitors-a network meta-analysis
Abstract
Background: Molecular events that drive endometriosis (EM) and cause accompanying immune deregulation remain elusive. Our purpose was to identify key pathways involved in lesion formation across diverse populations and to detect transcriptomic changes in eutopic endometrium that accompany EM.
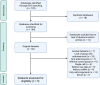
Methods: We searched Gene Expression Omnibus and ArrayExpress and performed differential gene expression analysis and a network meta-analysis on nine qualifying datasets. Those contained transcriptomic data on 114 ectopic endometrium samples (EL), 138 eutopic endometrium samples from women with endometriosis (EEM), and 79 eutopic endometrium samples from women without endometriosis (EH). Gene ontology and enrichment analysis were performed in DAVID, Metascape, and Cytoscape, and drug repurposing was done in CMap.
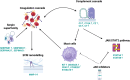
Results: EEM compared to EH upregulated CCL21 and downregulated BIRC3, CEL, and LEFTY1 genes (|log2FC| > 0.5, p < 0.05). EL showed increased expression of complement and serpin genes (EL vs. EEM: C7, logFC = 3.38, p < 0.0001; C3, logFC = 2.40, p < 0.0001; SERPINE1, logFC = 1.02, p < 0.05; SERPINE2, logFC = 1.54, p < 0.001) and mast cell markers (EL vs. EEM: CPA3, logFC = 1.54, p < 0.0001; KIT, logFC = 0.74, p < 0.001). Functional enrichment analysis highlighted complement and coagulation, inflammation, angiogenesis, and extracellular matrix remodeling as drivers of endometriosis. Pharmacogenomic analysis indicated Janus kinase (JAK), cyclin-dependent kinase (CDK), and topoisomerase inhibitors as therapy targets.
Conclusion: Our results suggest an interplay between complement and coagulation, mast cells, extracellular matrix remodeling, and the JAK/STAT3 pathway in endometriosis. We underscore the significance of complement C3 and propose JAK inhibitors as therapy candidates. Detected expression differences between EEM and EH are important for the development of diagnosis via endometrial biopsy.
Keywords: Janus kinase (JAK) inhibitors; complement and coagulation; endometriosis; eutopic and ectopic endometrium; mast cells; network meta-analysis.
Copyright © 2025 Golinska, Rycerz, Sobczak, Chrzanowski, Stawiski and Fendler.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
-
- Nnoaham KE, Hummelshoj L, Webster P, D’Hooghe T, De Cicco Nardone F, De Cicco Nardone C, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertility Sterility. (2011) 96:366–73.e8. doi: 10.1016/j.fertnstert.2011.05.090, PMID: - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

