Critical Role of IL1R2-ENO1 Interaction in Inhibiting Glycolysis-Mediated Pyroptosis for Protection Against Lethal Sepsis
- PMID: 40704655
- PMCID: PMC12533375
- DOI: 10.1002/advs.202502297
Critical Role of IL1R2-ENO1 Interaction in Inhibiting Glycolysis-Mediated Pyroptosis for Protection Against Lethal Sepsis
Abstract
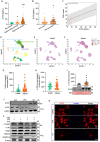
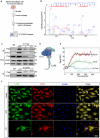
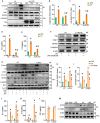
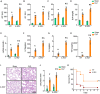
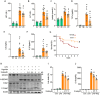
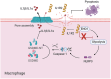
Immune cell metabolic reprogramming toward glycolysis is vital for sepsis defense. While interleukin 1 receptor 2 (IL1R2) acts as a decoy receptor for IL1α/β, its potential impact on cell metabolism and death during sepsis remains unclear. This study observed elevated plasma soluble IL1R2 (sIL1R2) levels in septic patients and mice. In pyroptotic macrophages, reduced intracellular IL1R2 expression led to its release extracellularly. Proteomic screening identified enolase 1 (ENO1), a key glycolysis enzyme, as the binding partner of IL1R2 in macrophages. IL1R2 suppresses ENO1 activity to inhibit glycolysis, gasdermin D (GSDMD)-mediated pyroptosis, and inflammation in macrophages. IL1R2-deficient mice exhibited heightened susceptibility to sepsis, with increased inflammation, organ injury, and mortality. Notably, ENO1 inhibition reduced inflammation, organ injury, and improved survival rates in septic mice. The study reveals that IL1R2 interacts with ENO1 to inhibit glycolysis-mediated pyroptosis and inflammation in sepsis, suggesting the IL1R2-ENO1 interaction as a promising therapeutic target of sepsis.
Keywords: IL1R2; enolase 1; glycolysis; macrophages; pyroptosis; sepsis.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Singer M., Deutschman C. S., Seymour C. W., Shankar‐Hari M., Annane D., Bauer M., Bellomo R., Bernard G. R., Chiche J. D., Coopersmith C. M., Hotchkiss R. S., Levy M. M., Marshall J. C., Martin G. S., Opal S. M., Rubenfeld G. D., van der Poll T., Vincent J. L., Angus D. C., JAMA, J. Am. Med. Assoc. 2016, 315, 801. - PMC - PubMed
-
- a) Latz E., Xiao T. S., Stutz A., Nat. Rev. Immunol. 2013, 13, 397; - PMC - PubMed
- b) Kayagaki N., Stowe I. B., Lee B. L., O'Rourke K., Anderson K., Warming S., Cuellar T., Haley B., Roose‐Girma M., Phung Q. T., Liu P. S., Lill J. R., Li H., Wu J., Kummerfeld S., Zhang J., Lee W. P., Snipas S. J., Salvesen G. S., Morris L. X., Fitzgerald L., Zhang Y., Bertram E. M., Goodnow C. C., Dixit V. M., Nature 2015, 526, 666; - PubMed
- c) Shi J., Zhao Y., Wang K., Shi X., Wang Y., Huang H., Zhuang Y., Cai T., Wang F., Shao F., Nature 2015, 526, 660; - PubMed
- d) Denning N. L., Aziz M., Gurien S. D., Wang P., Front. Immunol. 2019, 10, 2536. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
