HDACs and Their Inhibitors on Post-Translational Modifications: The Regulation of Cardiovascular Disease
- PMID: 40710369
- PMCID: PMC12293319
- DOI: 10.3390/cells14141116
HDACs and Their Inhibitors on Post-Translational Modifications: The Regulation of Cardiovascular Disease
Abstract
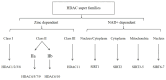
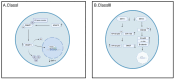
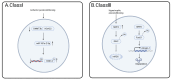
Cardiovascular diseases (CVD), such as myocardial hypertrophy, heart failure, atherosclerosis, and myocardial ischemia/reperfusion (I/R) injury, are among the major threats to human health worldwide. Post-translational modifications alter the function of proteins through dynamic chemical modification after synthesis. This mechanism not only plays an important role in maintaining homeostasis and plays a crucial role in maintaining normal cardiovascular function, but is also closely related to the pathological state of various diseases. Histone deacetylases (HDACs) play an important role in the epigenetic regulation of gene expression, and play important roles in post-translational modification by catalyzing the deacetylation of key lysine residues in nucleosomal histones, which are closely associated with the occurrence and development of cardiovascular diseases. Recent studies indicate that HDAC inhibitors (HDACis) may represent a new class of drugs for the treatment of cardiovascular diseases by influencing post-translational modifications. In this review, we systematically summarize the mechanism of action of HDACs and HDACis in post-translational modifications related to common cardiovascular diseases, providing new ideas for the treatment of CVD, and explore possible future research directions on the relationship between HDAC and HDACi in post-translational modifications and cardiovascular diseases.
Keywords: HDAC; HDACi; cardiovascular disease (CVD); post-translational modification (PTM).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Chemical Versatility in Catalysis and Inhibition of the Class IIb Histone Deacetylases.Acc Chem Res. 2024 Apr 16;57(8):1135-1148. doi: 10.1021/acs.accounts.3c00801. Epub 2024 Mar 26. Acc Chem Res. 2024. PMID: 38530703 Free PMC article. Review.
-
Unlocking the potential of histone deacetylase inhibitors in combatting age-related retinal degenerative diseases.Ageing Res Rev. 2025 Aug;110:102788. doi: 10.1016/j.arr.2025.102788. Epub 2025 May 31. Ageing Res Rev. 2025. PMID: 40456451 Review.
-
Contribution of histone deacetylases (HDACs) to the regulation of histone and non-histone proteins: implications for fibrotic diseases.BMB Rep. 2025 Jul 10:6322. Online ahead of print. BMB Rep. 2025. PMID: 40635201
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Effects of a gluten-reduced or gluten-free diet for the primary prevention of cardiovascular disease.Cochrane Database Syst Rev. 2022 Feb 24;2(2):CD013556. doi: 10.1002/14651858.CD013556.pub2. Cochrane Database Syst Rev. 2022. PMID: 35199850 Free PMC article.
References
-
- Malhotra A., Redberg R.F., Meier P. Saturated fat does not clog the arteries: Coronary heart disease is a chronic inflammatory condition, the risk of which can be effectively reduced from healthy lifestyle interventions. Br. J. Sports Med. 2017;51:1111–1112. doi: 10.1136/bjsports-2016-097285. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

