Metabolomic Profiling and Anti- Helicobacter pylori Activity of Caulerpa lentillifera (Sea Grape) Extract
- PMID: 40710507
- PMCID: PMC12298704
- DOI: 10.3390/md23070282
Metabolomic Profiling and Anti- Helicobacter pylori Activity of Caulerpa lentillifera (Sea Grape) Extract
Abstract
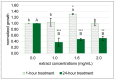
Helicobacter pylori is a gastric pathogen implicated in peptic ulcer disease and gastric cancer. The increasing prevalence of antibiotic-resistant strains underscores the urgent need for alternative therapeutic strategies. In this study, we investigated the chemical composition and antibacterial activity of an aqueous extract from Caulerpa lentillifera (sea grape), a farm-cultivated edible green seaweed collected from Krabi Province, Thailand. Ultra-high-performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS) revealed that the extract was enriched in bioactive nucleosides and phenolic compounds. In vitro assays demonstrated dose-dependent inhibition of H. pylori growth following exposure to sea grape extract. Furthermore, untargeted intracellular metabolomic profiling of H. pylori cells treated with the extract uncovered significant perturbations in central carbon and nitrogen metabolism, including pathways associated with the tricarboxylic acid (TCA) cycle, one-carbon metabolism, and alanine, aspartate, and glutamate metabolism. Pyrimidine biosynthesis was selectively upregulated, indicating a potential stress-induced shift toward nucleotide salvage and DNA repair. Of particular note, succinate levels were markedly reduced despite accumulation of other TCA intermediates, suggesting disruption of electron transport-linked respiration. These findings suggest that bioactive metabolites from C. lentillifera impair essential metabolic processes in H. pylori, highlighting its potential as a natural source of antimicrobial agents targeting bacterial physiology.
Keywords: Caulerpa lentillifera; Helicobacter pylori; chemical composition; metabolomics; sea grape.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





Similar articles
-
Isolation of Protein and Peptides from Symbiont Bacteria of Green Algae, Caulerpa lentillifera and Their Potency as Anticancer.Asian Pac J Cancer Prev. 2025 Mar 1;26(3):767-774. doi: 10.31557/APJCP.2025.26.3.767. Asian Pac J Cancer Prev. 2025. PMID: 40156392 Free PMC article.
-
Helile formula from "Tai ping sheng hui fang": anti-Helicobacter pylori activity, gut microbiota modulation, and inflammatory response regulation.Front Cell Infect Microbiol. 2025 Jul 22;15:1603128. doi: 10.3389/fcimb.2025.1603128. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40766846 Free PMC article.
-
Colon cancer inhibitory properties of Caulerpa lentillifera polysaccharide and its molecular mechanisms based on three-dimensional cell culture model.Int J Biol Macromol. 2024 May;267(Pt 1):131574. doi: 10.1016/j.ijbiomac.2024.131574. Epub 2024 Apr 12. Int J Biol Macromol. 2024. PMID: 38615857
-
Eradication therapy for peptic ulcer disease in Helicobacter pylori-positive people.Cochrane Database Syst Rev. 2016 Apr 19;4(4):CD003840. doi: 10.1002/14651858.CD003840.pub5. Cochrane Database Syst Rev. 2016. PMID: 27092708 Free PMC article.
-
Non-invasive diagnostic tests for Helicobacter pylori infection.Cochrane Database Syst Rev. 2018 Mar 15;3(3):CD012080. doi: 10.1002/14651858.CD012080.pub2. Cochrane Database Syst Rev. 2018. PMID: 29543326 Free PMC article.
References
-
- Chen Y.-C., Malfertheiner P., Yu H.-T., Kuo C.-L., Chang Y.-Y., Meng F.-T., Wu Y.-X., Hsiao J.-L., Chen M.-J., Lin K.-P. Global prevalence of Helicobacter pylori infection and incidence of gastric cancer between 1980 and 2022. Gastroenterology. 2024;166:605–619. doi: 10.1053/j.gastro.2023.12.022. - DOI - PubMed
-
- Essaidi I., Bounder G., Jouimyi R.M., Boura H., Elyounsi I., Kheir F.-Z., Benomar H., Badre W., Zerouali K., Maachi F. Comparative study of Helicobacter pylori resistance to clarithromycin and metronidazole and its association with epidemiological factors in a moroccan population. Asian Pac. J. Cancer Prev. 2022;23:2755. doi: 10.31557/APJCP.2022.23.8.2755. - DOI - PMC - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

