Biofilm Formation, Antibiotic Resistance, and Virulence Analysis of Human and Avian Origin Klebsiella pneumoniae from Jiangsu, China
- PMID: 40711288
- PMCID: PMC12299882
- DOI: 10.3390/vetsci12070628
Biofilm Formation, Antibiotic Resistance, and Virulence Analysis of Human and Avian Origin Klebsiella pneumoniae from Jiangsu, China
Abstract
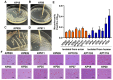
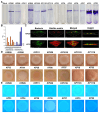
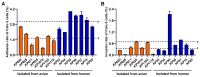
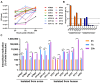
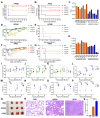
Klebsiella pneumoniae, a zoonotic pathogen of global concern, poses significant threats to both veterinary and public health. Here, a comparative study characterized 14 clinical isolates (7 avian-derived, 7 human-derived) from Jiangsu, China, through integrated genomic and phenotypic analyses. Firstly, multilocus sequence typing (MLST) revealed distinct epidemiological patterns: the same ST type in avian isolates was circulating between different species and different regions, whereas it was not found in human isolates. In addition, hypervirulent Klebsiella pneumoniae (hvKP) phenotypes confirmed by string test were exclusive to two human isolates (KP15, KP20). Secondly, biofilm detection demonstrated 78.6% (11/14) of isolates possessed biofilm-forming capacity, with cellulose but not curli as the predominant matrix component. Human-derived KP15 and KP20 had the strongest biofilm formation ability in all isolates. Antimicrobial susceptibility profiling identified serious multidrug resistance in both avian and human isolates. Virulence gene analysis revealed striking disparities, with human isolates harboring 10-20 virulence factors (median 15) versus 6-7 (median 6.5) in avian counterparts. Finally, functional pathogenesis assessments demonstrated human-derived strains exhibited stronger epithelial cell adhesion (2-fold higher) and invasion (1.97-fold higher) in Calu-3 cell models and paradoxically showed reduced macrophage phagocytosis (2.85-fold lower at 2 h) for immune escape. In vivo models confirmed dose-dependent mortality, with human isolates demonstrating higher lethality in both Galleria mellonella and mice. Virulence gene burden positively correlated with mortality outcomes. These findings delineate critical host adaptation differences in Klebsiella pneumoniae populations and provide empirical evidence for pathogen transmission dynamics at the human-animal interface.
Keywords: Klebsiella pneumoniae; antibiotic resistance; avian-origin; biofilm; human-origin; virulence.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures

References
Grants and funding
- 32172942/National Natural Science Foundation of China
- BE2022774/Key Research and Development Program of Social Development of Jiangsu Province
- CX(23)3071/Agricultural Science and Technology Independent Innovation Fund of Jiangsu Province
- BK20240045/Jiangsu Provincial Natural Science Fund for Distinguished Young Scholars
- LYKJ[2024]16/Forestry Science and Technology Innovation and Promotion Project of Jiangsu Province
LinkOut - more resources
Full Text Sources

