Malaria bivalent viral vectored vaccine protects against Plasmodium falciparum and vivax and blocks parasite transmission
- PMID: 40715105
- PMCID: PMC12297270
- DOI: 10.1038/s41541-025-01229-3
Malaria bivalent viral vectored vaccine protects against Plasmodium falciparum and vivax and blocks parasite transmission
Abstract
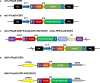
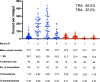
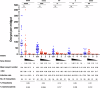
Malaria remains a major infectious disease, with Plasmodium falciparum and Plasmodium vivax often co-endemic, requiring a dual-target vaccine for adequate control. We previously developed monovalent vaccines against P. falciparum or P. vivax using vaccinia virus LC16m8Δ (m8Δ) and adeno-associated virus type 1 (AAV1). Here, we demonstrate the efficacy of a novel bivalent malaria vaccine against P. falciparum and P. vivax. The m8Δ vaccine harbors two gene cassettes encoding Pfs25-PfCSP and Pvs25-PvCSP fusion proteins, while the AAV1 vaccine includes two recombinant AAV1s carrying one of these cassettes as a mixture. Heterologous m8Δ-prime and AAV1-boost immunization provided 70% protection against both PfCSP/Pb and PvCSP/Pb transgenic sporozoites. Moreover, a membrane feeding assay using P. vivax isolates from infected patients in the Brazilian Amazon showed 90% transmission-blocking efficacy. The bivalent vaccine outperformed monovalent combinations, maintaining immune responses for over 7 months, and shows promise for malaria control and elimination.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: Authors S.Y., H.S., H.M., and M.I. are credited as inventors of patents concerning viral-vectored malaria vaccines 2022-24221. HS is also credited as an inventor on a pending patent related to LC16m8Δ WO 2005/054451 A1. However, neither of these products has been brought to market. The remaining authors declare that the research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
A two-dose viral-vectored Plasmodium vivax multistage vaccine confers durable protection and transmission-blockade in a pre-clinical study.Front Immunol. 2024 Apr 30;15:1372584. doi: 10.3389/fimmu.2024.1372584. eCollection 2024. Front Immunol. 2024. PMID: 38745665 Free PMC article.
-
Adeno-associated virus-based malaria booster vaccine following attenuated replication-competent vaccinia virus LC16m8Δ priming.Parasitol Int. 2023 Feb;92:102652. doi: 10.1016/j.parint.2022.102652. Epub 2022 Aug 23. Parasitol Int. 2023. PMID: 36007703
-
Sterile protection and transmission blockade by a multistage anti-malarial vaccine in the pre-clinical study.Front Immunol. 2022 Sep 29;13:1005476. doi: 10.3389/fimmu.2022.1005476. eCollection 2022. Front Immunol. 2022. PMID: 36248835 Free PMC article.
-
WITHDRAWN: Vaccines for preventing malaria.Cochrane Database Syst Rev. 2007 Jul 18;2003(4):CD000129. doi: 10.1002/14651858.CD000129.pub2. Cochrane Database Syst Rev. 2007. PMID: 17636596 Free PMC article.
-
Vaccines for preventing malaria.Cochrane Database Syst Rev. 2003;(1):CD000129. doi: 10.1002/14651858.CD000129. Cochrane Database Syst Rev. 2003. Update in: Cochrane Database Syst Rev. 2007 Jul 18;(4):CD000129. doi: 10.1002/14651858.CD000129.pub2. PMID: 12535387 Updated.
References
-
- Bourzac, K. Infectious disease: beating the big three. Nature507, S4–S7 (2014). - PubMed
-
- Venkatesan, P. WHO World Malaria Report 2024. Lancet Microbe, 101073 10.1016/j.lanmic.2025.101073 (2025). - PubMed
-
- Mendis, K., Sina, B. J., Marchesini, P. & Carter, R. The neglected burden of Plasmodium vivax malaria. Am. J. Trop. Med. Hyg.64, 97–106 (2001). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

