Auto-adjusting positive airway pressure: the fine line between engineering and medicine
- PMID: 40719998
- PMCID: PMC12304072
- DOI: 10.1007/s11325-025-03409-w
Auto-adjusting positive airway pressure: the fine line between engineering and medicine
Abstract
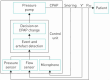
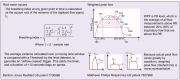
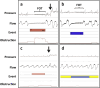
Auto-adjusting positive airway pressure (APAP), unlike continuous PAP (CPAP), dynamically adjusts treatment pressure in response to events detected automatically from a derived flow signal. Introduced in the 90's, APAP quickly became a key tool in sleep clinics, initially serving as a faster alternative to manual titration for patients with obstructive sleep apnea (OSA), and later also as a long-term treatment option to expedite follow-up visits. APAP and CPAP are overall comparable in terms of adherence, efficacy and control of symptoms. However, concern remains that APAP offers less control of chronic health outcomes, such as blood pressure, kidney function and glycemic values. Other APAP-related challenges entail engineering aspects. A major issue is that APAP algorithms-which govern event detection/identification and pressure adjustments-are proprietary of and vary among manufacturers, making them poorly understood by clinicians. Furthermore, APAP algorithms do not always match-up well when compared to both manual titration or manually scored polysomnography, particularly in the presence of unintentional leak. Variability in event detection, leak compensation, and pressure adjustment algorithms among devices adds another layer of complexity to clinical decision-making. All this complicates the management of OSA patients, who could be left with substantial residual disordered breathing, high leak, and a wide pressure range.This review aims to bridge the gap between the clinical and engineering perspectives of APAP, providing an up-to-date overview of current knowledge and existing challenges that sleep clinicians should consider when managing OSA patients with PAP therapy.
Keywords: APAP; Algorithms; Automatic event detection; Continuous positive airway pressure; OSA therapy; Titration.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Research involving human participants and/or animals: No research involving human participants has been conducted related to this review. However, data about a preliminary investigation (so far unpublished elsewhere) where human participation was involved are reported. Informed consent: No research requiring an informed consent has been conducted related to this review. However, data about a preliminary investigation (so far unpublished elsewhere) where an informed consent was needed, and obtained, was involved are reported. Institution where work was performed: None, since no data collection was involved. Conflict of interest: LM received industry grants from Apnimed, Inc and Prosomnus independent of this work, and is consulting for SleepRes, Inc. as a medical director, and Apnimed, Inc. DPW received consultancy fee from Bairitone, Cerebra Health, Cryosa, Mosanna, Onera, Xtrodes, Apnimed, and SleepRes, Inc. WHN, MK and BH have ownership in SleepRes, Inc.
Figures






Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Evaluation and management of obstructive sleep apnea in adults hospitalized for medical care: an American Academy of Sleep Medicine clinical practice guideline.J Clin Sleep Med. 2025 Aug 21. doi: 10.5664/jcsm.11864. Online ahead of print. J Clin Sleep Med. 2025. PMID: 40838698
-
Practice parameters for the surgical modifications of the upper airway for obstructive sleep apnea in adults.Sleep. 2010 Oct;33(10):1408-13. doi: 10.1093/sleep/33.10.1408. Sleep. 2010. PMID: 21061864 Free PMC article.
-
Adherence to continuous positive airway pressure for the treatment of obstructive sleep apnea in neurodegenerative diseases: A systematic review.Sleep Med Rev. 2023 Oct;71:101836. doi: 10.1016/j.smrv.2023.101836. Epub 2023 Aug 9. Sleep Med Rev. 2023. PMID: 37586145
-
Educational, supportive and behavioural interventions to improve usage of continuous positive airway pressure machines in adults with obstructive sleep apnoea.Cochrane Database Syst Rev. 2014 Jan 8;(1):CD007736. doi: 10.1002/14651858.CD007736.pub2. Cochrane Database Syst Rev. 2014. Update in: Cochrane Database Syst Rev. 2020 Apr 7;4:CD007736. doi: 10.1002/14651858.CD007736.pub3. PMID: 24399660 Updated.
References
-
- Sullivan CE, Issa FG, Berthon-Jones M, Eves L (1981) Reversal of obstructive sleep Apnoea by continuous positive airway pressure applied through the Nares. Lancet 1(8225):862–865 - PubMed
-
- Messineo L, Hete B, Diesem R, Noah W (2024) Circuit-dependent carbon dioxide rebreathing during continuous positive airway pressure. Sleep Med 121:42–47 - PubMed
-
- Noah WH, White DP, Hete B, Messineo L (2025) Rebreathing during CPAP therapy and its implications in obstructive sleep apnea. J Clin Sleep Med - PubMed
-
- Hynes DJ, Mann DL, Landry SA, Joosten SA, Edwards BA, Hamilton GS (2024) Night to night variability in obstructive sleep apnea severity, the physiological endotypes and the frequency of flow limitation. Sleep - PubMed
-
- Polo O, Berthon-Jones M, Douglas NJ, Sullivan CE (1994) Management of obstructive sleep apnoea/hypopnoea syndrome. Lancet 344(8923):656–660 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

