Looking into Abnormal Co-Expressions of Tau and TDP-43 in the Realm of Mixed Dementia Types: A Double-Punch Scenario
- PMID: 40722308
- PMCID: PMC12293603
- DOI: 10.3390/brainsci15070716
Looking into Abnormal Co-Expressions of Tau and TDP-43 in the Realm of Mixed Dementia Types: A Double-Punch Scenario
Abstract
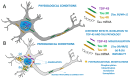
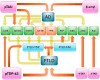
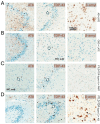
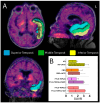
Transactive response DNA-binding protein of 43 kDa (TDP-43) and tau proteins play critical roles in neurodegenerative diseases, particularly frontotemporal lobar degeneration (FTLD) and Alzheimer's disease (AD). The co-occurrence of TDP-43 and tau pathologies raises questions about their role in disease progression. This review explores the simultaneous presence of tau and TDP-43 co-pathologies, emphasizing their molecular interactions and the resultant neuropathological implications. Additionally, we provide representative examples of their clinical presentations, neuroimaging, and neuropathological findings associated with FTLD-TDP and FTLD-tau, emphasizing the need for a comprehensive understanding of these intertwined pathologies. We analyze various clinical scenarios, including argyrophilic grain disease (AGD), primary age-related tauopathy (PART), and limbic predominant age-related TDP-43 encephalopathy (LATE), to elucidate the complex relationship between these proteinopathies. From the literature, the co-occurrence of tau and TDP-43 is linked to more severe and poorer clinical outcomes compared to isolated pathologies. This review underscores the necessity of considering co-pathologies in the context of FTLD, as they may act as accelerators of cognitive decline. This highlights the importance of integrated approaches in diagnosing and treating neurodegenerative conditions characterized by tau and TDP-43 misfolding. Understanding the interplay between these molecular markers is vital for advancing therapeutic strategies for such disorders.
Keywords: Alzheimer’s disease; TDP-43; biomarkers; frontotemporal lobar degeneration; molecular mechanisms; neuroimaging; neuropathology; tau.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Clinical criteria for a limbic-predominant amnestic neurodegenerative syndrome.Brain Commun. 2024 Jul 17;6(4):fcae183. doi: 10.1093/braincomms/fcae183. eCollection 2024. Brain Commun. 2024. PMID: 39021510 Free PMC article.
-
Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report.Brain. 2019 Jun 1;142(6):1503-1527. doi: 10.1093/brain/awz099. Brain. 2019. PMID: 31039256 Free PMC article.
-
Multimarker synaptic protein cerebrospinal fluid panels reflect TDP-43 pathology and cognitive performance in a pathological cohort of frontotemporal lobar degeneration.Mol Neurodegener. 2022 Apr 8;17(1):29. doi: 10.1186/s13024-022-00534-y. Mol Neurodegener. 2022. PMID: 35395770 Free PMC article.
-
Transactive response DNA-binding protein 43 (TDP-43) proteinopathy: the complex biological and clinical findings in limbic-predominant age-related TDP-43 encephalopathy (LATE) neuropathological changes, limbic-predominant amnestic neurodegenerative syndrome, and other mixed age-related major neurocognitive disorders.Curr Opin Psychiatry. 2025 Sep 1;38(5):341-347. doi: 10.1097/YCO.0000000000001025. Epub 2025 Jul 16. Curr Opin Psychiatry. 2025. PMID: 40709649 Review.
-
LATE-NC in Alzheimer's disease: Molecular aspects and synergies.Brain Pathol. 2024 Jul;34(4):e13213. doi: 10.1111/bpa.13213. Epub 2023 Oct 4. Brain Pathol. 2024. PMID: 37793659 Free PMC article. Review.
References
-
- Nelson P.T., Fardo D.W., Wu X., Aung K.Z., Cykowski M.D., Katsumata Y. Limbic-predominant age-related TDP-43 encephalopathy (LATE-NC): Co-pathologies and genetic risk factors provide clues about pathogenesis. J. Neuropathol. Exp. Neurol. 2024;83:396–415. doi: 10.1093/jnen/nlae032. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

