Effects of Betulinic Acid and Ursolic Acid on IL-17-Induced CCL20 Release in Normal Human Epidermal Keratinocytes
- PMID: 40724575
- PMCID: PMC12298970
- DOI: 10.3390/life15071073
Effects of Betulinic Acid and Ursolic Acid on IL-17-Induced CCL20 Release in Normal Human Epidermal Keratinocytes
Abstract
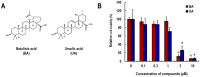
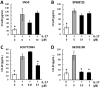
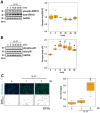
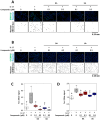
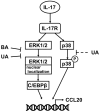
Psoriasis is a chronic inflammatory skin disease characterized by erythema, infiltration, and scaling, which is mainly caused by interleukin (IL)-17. The use of molecular targeted drugs in specific therapies offers high efficacy; however, high medical costs and a significant risk of side effects highlight the need for novel therapeutic agents. We previously observed that Morus alba extract (MAE) suppressed IL-17-induced CCL20 mRNA expression in normal human epidermal keratinocytes (NHEKs). In this study, we focused on the IL-17 signaling pathway and investigated the effects of pentacyclic triterpenoids, betulinic acid (BA), and ursolic acid (UA), which are present in MAE, on NHEK cells. Real-time reverse transcription polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) revealed that both BA and UA suppressed CCL20 expression, while only UA alone inhibited CCL20 release. ELISA using specific inhibitors demonstrated that both the p38 and extracellular-signal-regulated kinase 1/2 (ERK1/2) pathways were crucial for IL-17-induced CCL20 release in NHEK. UA effectively suppressed ERK1/2 nuclear localization and moderately affected p38 phosphorylation. These results indicated that UA is a potential seed compound for psoriasis treatment through its targeting of the IL-17 pathway.
Keywords: C-C motif chemokine ligand 20; betulinic acid; epidermal keratinocyte; interleukin 17; psoriasis; ursolic acid.
Conflict of interest statement
This study was conducted as part of a collaborative research project with Hinoki Shinyaku Co., Ltd., which provided financial support and was involved in the study design, data interpretation, and manuscript preparation. T. Oyama is an employee of Hinoki Shinyaku Co., Ltd. and was seconded to T. Hatanaka’s laboratory during the study. S. Abe is the president of the company. Although several reagents used in this study were provided by Hinoki Shinyaku Co., Ltd., they are not proprietary materials and are commercially available from multiple vendors. The authors affirm that all scientific analyses and conclusions were reached with due objectivity, and the interpretation of the data was carried out in accordance with academic standards.
Figures


Similar articles
-
Interleukin (IL)-34 promotes the inflammatory role of IL-1β-producing myeloid cells in pemphigus lesions.Br J Dermatol. 2025 Jul 17;193(2):287-297. doi: 10.1093/bjd/ljaf130. Br J Dermatol. 2025. PMID: 40203120
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2020 Jan 9;1(1):CD011535. doi: 10.1002/14651858.CD011535.pub3. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Apr 19;4:CD011535. doi: 10.1002/14651858.CD011535.pub4. PMID: 31917873 Free PMC article. Updated.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
-
- Nograles K.E., Zaba L.C., Guttman-Yassky E., Fuentes-Duculan J., Suárez-Fariñas M., Cardinale I., Khatcherian A., Gonzalez J., Pierson K.C., White T.R. Th17 cytokines interleukin (IL)-17 and IL-22 modulate distinct inflammatory and keratinocyte-response pathways. Br. J. Dermatol. 2008;159:1092–1102. doi: 10.1111/j.1365-2133.2008.08769.x. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

