Unlocking Implantation: The Role of Nitric Oxide, NO2-NO3, and eNOS in Endometrial Receptivity and IVF Success-A Systematic Review
- PMID: 40724818
- PMCID: PMC12295810
- DOI: 10.3390/ijms26146569
Unlocking Implantation: The Role of Nitric Oxide, NO2-NO3, and eNOS in Endometrial Receptivity and IVF Success-A Systematic Review
Abstract
Nitric oxide (NO) predominantly regulates endometrial receptivity, angiogenesis, immunological tolerance, and trophoblast invasion throughout the implantation period. Both insufficient and excessive nitric oxide production have been linked to suboptimal embryo implantation and infertility. The primary enzymatic source of uterine nitric oxide, along with hormonal, metabolic, and immunological variables and genetic variations in the endothelial nitric oxide synthase gene (NOS3), affects endothelial nitric oxide synthase (eNOS). Despite its considerable importance, there is limited knowledge regarding the practical implementation of nitric oxide-related diagnoses and therapies in reproductive medicine. A comprehensive assessment was performed in accordance with the PRISMA principles. Electronic searches were carried out in PubMed, Scopus, and Embase, and we analyzed the literature published from 2000 to 2024 regarding the association between NO, its metabolites (NO2- and NO3-), eNOS expression, NOS3 gene variants, and reproductive outcomes. Relevant studies encompassed clinical trials, observational studies, and experimental research using either human or animal subjects. We collected data about therapeutic interventions, hormonal and immunological associations, nitric oxide measurement techniques, and in vitro fertilization success rates. A total of thirty-four studies were included. Dysregulated nitric oxide signaling, characterized by modified eNOS expression, oxidative stress, or NOS3 polymorphisms (e.g., Glu298Asp and intron 4 VNTR), was linked to diminished endometrial receptivity and an elevated risk of implantation failure and miscarriage. The dynamics of local uterine NO are essential as elevated and diminished systemic levels of NO2-/NO3- corresponded with enhanced and decreased implantation rates, respectively. Among many therapeutic approaches, targeted hormone treatments, antioxidant therapy, and dietary nitrate supplements have demonstrated potential in restoring nitric oxide balance and enhancing reproductive outcomes. In animal models, the modification of nitric oxide significantly impacted decidualization, angiogenesis, and embryo viability. Nitric oxide is a multifaceted molecular mediator with considerable ramifications for successful implantation. Its therapeutic and diagnostic efficacy increases with its sensitivity to environmental, hormonal, and genetic alterations. Integrating targeted nitric oxide modulation, oxidative stress assessment, and NOS3 genotyping with personalized reproductive therapy will enhance endometrial receptivity and improve IVF outcomes. Future translational research should incorporate nitric oxide signaling into personalized treatment protocols for patients with unexplained infertility or recurrent implantation failure.
Keywords: NO2−; NO3−; NOS3 polymorphisms; eNOS; endometrial microenvironment; endometrial receptivity; implantation failure; in vitro fertilization (IVF); nitric oxide; recurrent pregnancy loss; unexplained infertility.
Conflict of interest statement
The authors declare no conflicts of interest.
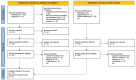
Figures
Similar articles
-
Podocalyxin is a key negative regulator of human endometrial epithelial receptivity for embryo implantation.Hum Reprod. 2021 Apr 20;36(5):1353-1366. doi: 10.1093/humrep/deab032. Hum Reprod. 2021. PMID: 33822049 Free PMC article.
-
Autologous platelet-rich plasma for assisted reproduction.Cochrane Database Syst Rev. 2024 Apr 29;4(4):CD013875. doi: 10.1002/14651858.CD013875.pub2. Cochrane Database Syst Rev. 2024. PMID: 38682756 Free PMC article.
-
Endometrial injury for pregnancy following sexual intercourse or intrauterine insemination.Cochrane Database Syst Rev. 2016 Jun 14;(6):CD011424. doi: 10.1002/14651858.CD011424.pub2. Cochrane Database Syst Rev. 2016. Update in: Cochrane Database Syst Rev. 2021 Mar 18;3:CD011424. doi: 10.1002/14651858.CD011424.pub3. PMID: 27296541 Updated.
-
In vitro fertilisation for unexplained subfertility.Cochrane Database Syst Rev. 2005 Apr 18;(2):CD003357. doi: 10.1002/14651858.CD003357.pub2. Cochrane Database Syst Rev. 2005. Update in: Cochrane Database Syst Rev. 2012 Apr 18;(4):CD003357. doi: 10.1002/14651858.CD003357.pub3. PMID: 15846658 Updated.
-
Endometrial injury to overcome recurrent embryo implantation failure: a systematic review and meta-analysis.Reprod Biomed Online. 2012 Dec;25(6):561-71. doi: 10.1016/j.rbmo.2012.08.005. Epub 2012 Sep 12. Reprod Biomed Online. 2012. PMID: 23063812
References
-
- Luo X., He Z., Ma R., Lin N., Li L., Li Y., Ke Y., Meng X., Wu Z. Narrative Review of Multifaceted Approaches to Managing Recurrent Implantation Failure: Insights and Innovations. Clin. Exp. Obstet. Gynecol. 2024;51:87. doi: 10.31083/j.ceog5104087. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

