Titin's Intrinsically Disordered PEVK Domain Modulates Actin Polymerization
- PMID: 40725251
- PMCID: PMC12295887
- DOI: 10.3390/ijms26147004
Titin's Intrinsically Disordered PEVK Domain Modulates Actin Polymerization
Abstract
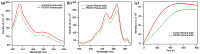
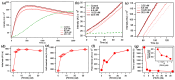
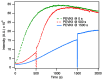
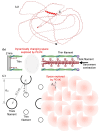
The multi-domain muscle protein titin provides elasticity and mechanosensing functions to the sarcomere. Titin's PEVK domain is intrinsically disordered due to the presence of a large number of prolines and highly charged residues. Although PEVK does not have canonical actin-binding motifs, it has been shown to bind F-actin. Here, we explored whether the PEVK domain may also affect actin assembly. We cloned the middle, 733-residue-long segment (called PEVKII) of the full-length PEVK domain, expressed in E. coli and purified by using His- and Avi-tags engineered to the N- and C-termini, respectively. Actin assembly was monitored by the pyrene assay in the presence of varying PEVKII concentrations. The structural features of PEVKII-associated F-actin were studied with atomic force microscopy. The added PEVKII enhanced the initial and log-phase rates of actin assembly and the peak F-actin quantity in a concentration-dependent way. However, the critical concentration of actin polymerization was unaltered. Thus, PEVK accelerates actin polymerization by facilitating its nucleation. This effect was highlighted in the AFM images of F-actin-PEVKII adsorbed to the supported lipid bilayer. The sample was dominated by radially symmetric complexes of short actin filaments. PEVK's actin polymerization-modulating effect may, in principle, have a function in regulating sarcomeric actin length and turnover. Altogether, titin's PEVK domain is not only a non-canonical actin-binding protein that regulates sarcomeric shortening, but one that may modulate actin polymerization as well.
Keywords: AlphaFold; PEVK; actin paracrystal; atomic force microscopy; intrinsically unstructured protein domain; polymerization assay; pyrene actin; supported lipid bilayer; titin.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Direction-dependent contributions of cardiac myofilament networks to myocardial passive stiffness reveal a major disparity for titin.Basic Res Cardiol. 2025 Aug;120(4):761-777. doi: 10.1007/s00395-025-01119-8. Epub 2025 Jun 13. Basic Res Cardiol. 2025. PMID: 40512239 Free PMC article.
-
Glycerol storage increases passive stiffness of muscle fibers through effects on titin extensibility.J Gen Physiol. 2025 Jul 7;157(4):e202413729. doi: 10.1085/jgp.202413729. Epub 2025 May 9. J Gen Physiol. 2025. PMID: 40341854 Free PMC article.
-
Structural dynamics of IDR interactions in human SFPQ and implications for liquid-liquid phase separation.Acta Crystallogr D Struct Biol. 2025 Jul 1;81(Pt 7):357-379. doi: 10.1107/S2059798325005303. Epub 2025 Jun 27. Acta Crystallogr D Struct Biol. 2025. PMID: 40574713 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

