Retrospective analysis of quality monitoring indexes of Grifols Panther nucleic acid detection system
- PMID: 40725892
- PMCID: PMC12303454
- DOI: 10.1097/MD.0000000000043463
Retrospective analysis of quality monitoring indexes of Grifols Panther nucleic acid detection system
Abstract
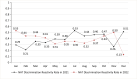
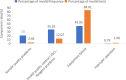
Nucleic acid amplification testing (NAT) is a highly sensitive and specific testing technology for bloodborne infectious diseases. Since 2010, blood collection and supply institutions in China have widely adopted NAT technology for blood screening, greatly enhancing blood safety. Basing it on comparison with established quality systems in serological testing processes, we aimed to establish and continuously improve a quality monitoring system that meets the requirements of NAT procedure. Statistical analysis was conducted using the daily performance data of the Grifols Procleix Panther nucleic acid testing system in the laboratory from 2021 to 2022, including test frequency reactive rate, invalid test rate, system failures frequency, and laboratory contamination monitoring indicators. In 2021 and 2022, the annual discriminated rate on NAT yield donation were 39.11% and 39.29%, respectively. The average annual invalid test rates for the 4 units of Grifols Panther systems were 1.80%, 1.47%, 1.60%, and 1.51%, resulting in an overall invalid test rate of 1.60% (7881/493, 137). A total of 377 invalid runs occurred, with the most frequent invalid test reasons being 64 instances of insufficient internal control detection ("low internal control"), 55 instances of issues with verifying the volume of target capture reagent + samples ("VVFS"), and 49 instances of the presence of clots detected during aspiration ("CLT"). The most significant contributor to invalid test counts was pipette malfunction ("PMFR"), which accounted for 2136 invalid tests. In the laboratory contamination monitoring, a total of 792 monitoring tests were conducted over a 2-year period, during which hepatitis B virus DNA was detected once in a specimen centrifuge cup. Establishing systematic quality monitoring indicators can effectively and promptly identify potential quality risk factors, thereby ensuring the quality of blood testing.
Keywords: Grifols Panther nucleic acid testing system; blood screening; quality control; quality monitoring indicators; voluntary blood donation.
Copyright © 2025 the Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures
References
-
- Faddy HM, Osiowy C, Custer B, et al. International review of blood donation nucleic acid amplification testing. Vox Sang. 2024;119:315–25. - PubMed
-
- Wang R, Xue X, Xiao Y, Liu Y, Yu Y. The prevalence of occult hepatitis B infection among the blood donors in a donation center in Beijing. Diagn Microbiol Infect Dis. 2024;109:116240. - PubMed
-
- Van de laar T, Hogema B, Molenaar-de backer MW, Marijt-van kreek T, Zaaijer HL. Blood donor screening in the Netherlands: universal anti-HBc screening in combination with HBV nucleic acid amplification testing may allow discontinuation of hepatitis B virus antigen testing. Transfusion. 2021;61:2116–24. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

