Camrelizumab, an Anti-PD-1 Monoclonal Antibody, Plus Carboplatin and Nab-Paclitaxel as First-Line Setting for Extensive-Stage Small-Cell Lung Cancer: A Phase 2 Trial and Biomarker Analysis
- PMID: 40727251
- PMCID: PMC12301171
- DOI: 10.1002/mco2.70300
Camrelizumab, an Anti-PD-1 Monoclonal Antibody, Plus Carboplatin and Nab-Paclitaxel as First-Line Setting for Extensive-Stage Small-Cell Lung Cancer: A Phase 2 Trial and Biomarker Analysis
Abstract
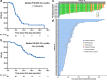
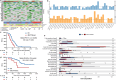
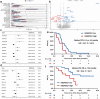
This study aimed to investigate the efficacy, safety, and predictors of camrelizumab combined with carboplatin and nab-paclitaxel as first-line setting for patients with extensive-stage small-cell lung cancer (ES-SCLC). Camrelizumab plus carboplatin and nab-paclitaxel were administrated every 3 weeks for four to six cycles, followed by maintenance camrelizumab until intolerable toxicity or disease progression. The primary endpoint was 6-month progression-free survival (PFS) rate and secondary endpoints were objective response rate (ORR), disease control rate (DCR), PFS, overall survival (OS), and safety. We conducted the whole-exome and transcriptomic sequencing on available tumor samples to explore the potential predictive biomarkers. A total of 60 patients were included. Primary endpoint was met with 6-month PFS rate of 52.2%. The median PFS and OS were 7.1 and 18.1 months, respectively. The confirmed ORR and DCR were 73.3% and 93.3%, respectively. No unexpected adverse events were observed. Exploratory analysis showed that MUC17 alterations or high NEUROG1 expression were correlated with markedly shorter PFS and OS. Deeper investigation of transcriptomic data reveals two subsets with distinct immune features and therapeutic vulnerabilities. Collectively, this trial suggested that camrelizumab plus carboplatin and nab-paclitaxel might be an alternative first-line setting for ES-SCLC. Integration of multiomic data could highlight the complex mechanisms underlying chemo-immunotherapy responses.
Keywords: ES‐SCLC; biomarker; camrelizumab; immunotherapy; nab‐paclitaxel.
© 2025 The Author(s). MedComm published by Sichuan International Medical Exchange & Promotion Association (SCIMEA) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
Shengxiang Ren has reported receiving speaker honoraria from Roche, Lilly, Boehringer Ingelheim, Hengrui, Merck Sharp & Dohme, and Junshi, and advisory fees from Hengrui, Merck Sharp & Dohme, and Roche. No further disclosures were reported. The other authors declare no conflicts of interest.
Figures


Similar articles
-
Phase II trial of nab-paclitaxel plus ramucirumab in combination with nivolumab for unresectable advanced or recurrent gastric cancer after progression on first-line treatment including fluoropyrimidine, platinum, and anti-PD-1/PD-L1 antibody (PADDLE).BMC Cancer. 2025 Feb 4;25(1):201. doi: 10.1186/s12885-025-13591-5. BMC Cancer. 2025. PMID: 39905373 Free PMC article.
-
Efficacy and safety of first-line PD-1/PD-L1 inhibitors combined with or without anti-angiogenesis therapy for extensive-stage small-cell lung cancer: a network meta-analysis.Ther Adv Med Oncol. 2025 Jun 25;17:17588359251348310. doi: 10.1177/17588359251348310. eCollection 2025. Ther Adv Med Oncol. 2025. PMID: 40574965 Free PMC article.
-
Interim analysis of the efficiency and safety of neoadjuvant PD-1 inhibitor (sintilimab) combined with chemotherapy (nab-paclitaxel and carboplatin) in potentially resectable stage IIIA/IIIB non-small cell lung cancer: a single-arm, phase 2 trial.J Cancer Res Clin Oncol. 2023 Feb;149(2):819-831. doi: 10.1007/s00432-021-03896-w. Epub 2022 Feb 22. J Cancer Res Clin Oncol. 2023. PMID: 35192053 Free PMC article. Clinical Trial.
-
Comparison of efficacy and safety of PD-1/PD-L1 combination therapy in first-line treatment of advanced NSCLC: an updated systematic review and network meta-analysis.Clin Transl Oncol. 2024 Oct;26(10):2488-2502. doi: 10.1007/s12094-024-03442-3. Epub 2024 Apr 16. Clin Transl Oncol. 2024. PMID: 38625495
-
Weekly versus tri-weekly paclitaxel with carboplatin for first-line treatment in women with epithelial ovarian cancer.Cochrane Database Syst Rev. 2022 Feb 21;2(2):CD012007. doi: 10.1002/14651858.CD012007.pub2. Cochrane Database Syst Rev. 2022. PMID: 35188221 Free PMC article.
References
-
- Simpson K. L., Rothwell D. G., Blackhall F., and Dive C., “Challenges of Small Cell Lung Cancer Heterogeneity and Phenotypic Plasticity,” Nature Reviews Cancer 25, no. 6 (2025): 447–462. - PubMed
-
- Sung H., Ferlay J., Siegel R. L., et al., “Global Cancer Statistics 2020: Globocan Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries,” CA: A Cancer Journal for Clinicians 71, no. 3 (2021): 209–249. - PubMed
-
- van Meerbeeck J. P., Fennell D. A., and De Ruysscher D. K., “Small‐Cell Lung Cancer,” Lancet 378, no. 9804 (2011): 1741–1755. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
