Higher p-Cresol sulfate and tumor necrosis factor (TNF)-α levels are associated with cognitive improvement through increased TNF receptor 2 in individuals with mild cognitive impairment: A cross-sectional mediation analysis
- PMID: 40727257
- PMCID: PMC12301611
- DOI: 10.1177/25424823251362483
Higher p-Cresol sulfate and tumor necrosis factor (TNF)-α levels are associated with cognitive improvement through increased TNF receptor 2 in individuals with mild cognitive impairment: A cross-sectional mediation analysis
Abstract
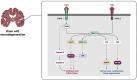
Background: Neuroinflammation in neurocognitive disorders is driven by the release of tumor necrosis factor (TNF)-α from brain immune cells in response to injury, infection, or p-Cresol sulfate (p-CS)-a metabolite associated with chronic kidney disease and linked to TNF-α activity. However, the underlying mechanisms through which TNF-α and p-CS influence cognitive performance remain unclear.
Objective: This study investigated the impact of TNF-α and p-CS on cognition, focusing on the role of TNF Receptor 2 (TNFR2) in cognitively normal individuals (CN; n = 36), Alzheimer's disease patients (AD; n = 85), and those with mild cognitive impairment (MCI; n = 219).
Methods: Cognitive status was assessed with ADAS-Cog 13, p-CS measured via MxP® Quant 500, and TNF-α/TNFR2 quantified using Human DiscoveryMAP®. Mediation analysis explored TNFR2's role in linking p-CS, TNF-α, and cognition, with significance set at p < 0.05 and FDR controlled by the Benjamini-Hochberg method.
Results: The results showed that TNF-α levels were slightly higher in AD than in MCI, while TNFR2 levels were lowest in MCI, higher in CN, and highest in AD. After adjusting for age, gender, and APOE ɛ3/ɛ4 status, higher TNF-α levels were associated with higher TNFR2 levels in both MCI and AD. In MCI, elevated TNFR2 correlated with better cognitive function, indicating a possible neuroprotective role at this stage of cognitive decline. Further analysis revealed that both p-CS and TNF-α contributed to increased TNFR2 levels, which in turn supported cognitive performance.
Conclusions: In short, p-CS and TNF-α may improve cognitive performance via TNFR2 in individuals with MCI.
Keywords: Alzheimer's disease; TNF receptor 2; cognitive performance; mild cognitive impairment; neuroinflammation; p-Cresyl sulfate; tumor necrosis factor-alpha.
© The Author(s) 2025.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
Similar articles
-
Vitamin E for Alzheimer's dementia and mild cognitive impairment.Cochrane Database Syst Rev. 2017 Apr 18;4(4):CD002854. doi: 10.1002/14651858.CD002854.pub5. Cochrane Database Syst Rev. 2017. PMID: 28418065 Free PMC article.
-
Vitamin E for Alzheimer's dementia and mild cognitive impairment.Cochrane Database Syst Rev. 2017 Jan 27;1(1):CD002854. doi: 10.1002/14651858.CD002854.pub4. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2017 Apr 18;4:CD002854. doi: 10.1002/14651858.CD002854.pub5. PMID: 28128435 Free PMC article. Updated.
-
Elevated CSF GAP-43 in Mild Cognitive Impairment Linked to Cognitive Impairment Through Increased Amyloid-β Accumulation, with a Shift to Reduced Amyloid-β Accumulation in Alzheimer's Disease.J Mol Neurosci. 2025 Mar 20;75(2):39. doi: 10.1007/s12031-025-02333-8. J Mol Neurosci. 2025. PMID: 40111590
-
CSF Amyloid-β42 associates with neuropsychiatric and cognitive outcomes via cerebral glucose metabolism.Mol Brain. 2025 Jul 1;18(1):55. doi: 10.1186/s13041-025-01229-3. Mol Brain. 2025. PMID: 40597399 Free PMC article.
-
Cholinesterase inhibitors for vascular dementia and other vascular cognitive impairments: a network meta-analysis.Cochrane Database Syst Rev. 2021 Feb 22;2(2):CD013306. doi: 10.1002/14651858.CD013306.pub2. Cochrane Database Syst Rev. 2021. PMID: 33704781 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

