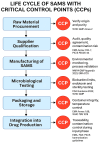
Preventing Microorganism Contamination in Starting Active Materials for Synthesis from Global Regulatory Agencies: Overview for Public Health Implications
- PMID: 40732104
- PMCID: PMC12298561
- DOI: 10.3390/microorganisms13071595
Preventing Microorganism Contamination in Starting Active Materials for Synthesis from Global Regulatory Agencies: Overview for Public Health Implications
Abstract
Starting Active Materials for Synthesis (SAMS) represents a critical stage in drug manufacturing, directly influencing the microbiological quality and safety of the final product. The introduction of SAMS marks the point where Good Manufacturing Practices (GMP) begin to apply, which are essential for ensuring sterility and preventing microbial contamination during the synthesis process. However, defining the exact point in the process that qualifies as the SAMS is subject to uncertainties, as earlier stages are not always governed by stringent GMP standards. The regulatory differences between various countries further contribute to this issue. This study explores the implications of SAMS selection and use in relation to sterility and infection control, analyzing the guidelines of major Regulatory Authorities and comparing their approaches to GMP. Regulations from several international regulatory agencies were examined, with a particular focus on microbiological control measures and infection protection in the SAMS manufacturing process. The analysis focused on the microbiological control requirements and safety measures applicable to the stages preceding the introduction of SAMS into the production of the final Active Pharmaceutical Ingredients (APIs). Documents published between 2015 and 2025 were included based on predefined criteria regarding relevance, accessibility, and regulatory authority. The analysis revealed significant discrepancies between regulations regarding the definition and management of SAMS. In particular, the regulations in Mexico and India have notable gaps, failing to provide clear guidelines on SAMS sterility and protection against infectious contamination. Conversely, China has introduced risk-based approaches and early-stage microbiological controls, especially for sterile products, aligning with international standards. The European Medicines Agency (EMA), the U.S. Food and Drug Administration (FDA), the Pharmaceutical Inspection Co-operation Scheme (PIC/S), and the World Health Organization (WHO) have well-established systems for microbiological quality control of SAMS, including rigorous measures for the validation of suppliers and risk management to ensure that SAMS does not compromise the microbiological safety of the final product. The regulations in Brazil and Canada introduce additional measures to protect the microbiological quality of SAMS, with specifications for contamination control and certification of critical stages. The lack of a harmonized language for the definition of SAMS, coupled with a fragmented regulatory framework, presents a challenge for infection protection in pharmaceutical manufacturing. Key issues include the absence of specific regulations for stages prior to the introduction of SAMS and the lack of standards for inspections related to these stages. A desirable solution would be the mandatory extension of GMPs to the stages before SAMS introduction, with centralized control to ensure sterility and protection against infection throughout the entire manufacturing process.
Keywords: global regulatory agencies; overview; preventing microorganism contamination; public health; starting active materials for synthesis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Is Your Surgical Helmet System Compromising the Sterile Field? A Systematic Review of Contamination Risks and Preventive Measures in Total Joint Arthroplasty.Clin Orthop Relat Res. 2025 Jun 1;483(6):972-990. doi: 10.1097/CORR.0000000000003383. Epub 2025 Feb 5. Clin Orthop Relat Res. 2025. PMID: 39915114
-
[Volume and health outcomes: evidence from systematic reviews and from evaluation of Italian hospital data].Epidemiol Prev. 2013 Mar-Jun;37(2-3 Suppl 2):1-100. Epidemiol Prev. 2013. PMID: 23851286 Italian.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2020 Jan 9;1(1):CD011535. doi: 10.1002/14651858.CD011535.pub3. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Apr 19;4:CD011535. doi: 10.1002/14651858.CD011535.pub4. PMID: 31917873 Free PMC article. Updated.
-
Health professionals' experience of teamwork education in acute hospital settings: a systematic review of qualitative literature.JBI Database System Rev Implement Rep. 2016 Apr;14(4):96-137. doi: 10.11124/JBISRIR-2016-1843. JBI Database System Rev Implement Rep. 2016. PMID: 27532314
References
-
- World Health Organization (WHO) WHO Guidelines on Good Manufacturing Practices for Active Pharmaceutical Ingredients. World Health Organization (WHO); Geneva, Switzerland: 2010. [(accessed on 25 April 2025)]. TRS No. 957. Available online: https://www.who.int/teams/health-product-policy-and-standards/standards-....
-
- European Medicines Agency (EMA) Guideline on the Requirements for Quality Documentation Concerning Biological Investigational Medicinal Products in Clinical Trials. European Medicines Agency (EMA); Amsterdam, The Netherlands: 2022. [(accessed on 25 April 2025)]. Available online: https://www.ema.europa.eu/en/requirements-quality-documentation-concerni....
-
- Asif M., Usmani S. Basics of Pharmaceutical Manufacturing and Quality Operations. CRC Press; Boca Raton, FL, USA: 2023. - DOI
-
- U.S. Food and Drug Administration (FDA) Questions and Answers on Current Good Manufacturing Practice—Control of Components and Drug Product Containers and Closures. U.S. Food and Drug Administration (FDA); Silver Spring, MD, USA: 2005. [(accessed on 24 June 2025)]. Question 3: Media fill contamination. Available online: https://www.fda.gov/drugs/guidances-drugs/questions-and-answers-current-....
Publication types
LinkOut - more resources
Full Text Sources
Research Materials