The Therapeutic Potential of Butyrate and Lauric Acid in Modulating Glial and Neuronal Activity in Alzheimer's Disease
- PMID: 40732911
- PMCID: PMC12298293
- DOI: 10.3390/nu17142286
The Therapeutic Potential of Butyrate and Lauric Acid in Modulating Glial and Neuronal Activity in Alzheimer's Disease
Abstract
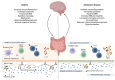
Alzheimer's disease (AD) is a progressive neurodegenerative disorder marked by amyloid-β plaque accumulation, tau tangles, and extensive neuroinflammation. Neuroinflammation, driven by glial cells like microglia and astrocytes, plays a critical role in AD progression. Initially, these cells provide protective functions, such as debris clearance and neurotrophic support. However, as AD progresses, chronic activation of these cells exacerbates inflammation, contributing to synaptic dysfunction, neuronal loss, and cognitive decline. Microglia release pro-inflammatory cytokines and reactive oxygen species (ROS), while astrocytes undergo reactive astrogliosis, further impairing neuronal health. This maladaptive response from glial cells significantly accelerates disease pathology. Current AD treatments primarily aim at symptomatic relief, with limited success in disease modification. While amyloid-targeting therapies like Aducanumab and Lecanemab show some promise, their efficacy remains limited. In this context, natural compounds have gained attention for their potential to modulate neuroinflammation and promote neuroprotection. Among these, butyrate and lauric acid are particularly notable. Butyrate, produced by a healthy gut microbiome, acts as a histone deacetylase (HDAC) inhibitor, reducing pro-inflammatory cytokines and supporting neuronal health. Lauric acid, on the other hand, enhances mitochondrial function, reduces oxidative stress, and modulates inflammatory pathways, thereby supporting glial and neuronal health. Both compounds have been shown to decrease amyloid-β deposition, reduce neuroinflammation, and promote neuroprotection in AD models. This review explores the mechanisms through which butyrate and lauric acid modulate glial and neuronal activity, highlighting their potential as therapeutic agents for mitigating neuroinflammation and slowing AD progression.
Keywords: Alzheimer’s disease; astrocytes; butyrate; lauric acid; microglia; neurons.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Unveiling the Protective Roles of Melatonin on Glial Cells in the Battle Against Alzheimer's Disease-Insights from In Vivo and In Vitro Studies.Mol Neurobiol. 2025 Aug;62(8):10414-10426. doi: 10.1007/s12035-025-04904-7. Epub 2025 Apr 10. Mol Neurobiol. 2025. PMID: 40208552 Review.
-
Microglial activation states and their implications for Alzheimer's Disease.J Prev Alzheimers Dis. 2025 Jan;12(1):100013. doi: 10.1016/j.tjpad.2024.100013. Epub 2025 Jan 1. J Prev Alzheimers Dis. 2025. PMID: 39800461 Free PMC article. Review.
-
Co-Aggregation of Syndecan-3 with β-Amyloid Aggravates Neuroinflammation and Cognitive Impairment in 5×FAD Mice.Int J Mol Sci. 2025 Jun 8;26(12):5502. doi: 10.3390/ijms26125502. Int J Mol Sci. 2025. PMID: 40564963 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Microglial activation as a hallmark of neuroinflammation in Alzheimer's disease.Metab Brain Dis. 2025 May 17;40(5):207. doi: 10.1007/s11011-025-01631-9. Metab Brain Dis. 2025. PMID: 40381069 Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

