A Semi-Mechanistic Mathematical Model of Immune Tolerance Induction to Support Preclinical Studies of Human Monoclonal Antibodies in Rats
- PMID: 40733054
- PMCID: PMC12300661
- DOI: 10.3390/pharmaceutics17070845
A Semi-Mechanistic Mathematical Model of Immune Tolerance Induction to Support Preclinical Studies of Human Monoclonal Antibodies in Rats
Abstract
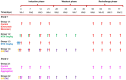
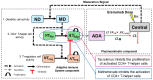
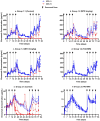
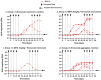
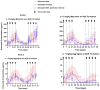
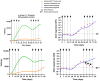
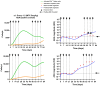
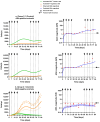
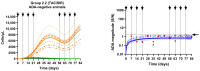
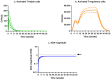
Background/Objectives: The administration of human monoclonal antibodies (mAb) in preclinical pharmacokinetics and toxicology studies often triggers an immune response, leading to the formation of anti-drug antibodies (ADA). To mitigate this effect, we have recently performed and reported on studies using short-term immunosuppressive regimens to induce prolonged immune tolerance towards a human mAb, erenumab, in rats. Here, we report on the development of a semi-mechanistic modeling approach that quantitatively integrates pharmacokinetic and immunogenicity assessments from immune tolerance induction studies to provide a framework for the simulation-based evaluation of different immune induction scenarios for the maintenance of prolonged immune tolerance towards human mAbs. Methods: The integrated pharmacokinetic/pharmacodynamic (PK/PD) modeling approach combined a semi-mechanistic model of the adaptive immune system to predict ADA formation kinetics with a population pharmacokinetic model to assess the impact of the time course of the ADA magnitude on the PK of erenumab in rats. Model-derived erenumab concentration-time profiles served as input for a quantitative system pharmacology-style semi-mechanistic model of the adaptive immune system to conceptualize the ADA response as a function of the kinetics of CD4+ T helper cells and T regulatory cells. Results: The model adequately described the observed ADA magnitude-time profiles in all treatment groups and reasonably simulated the kinetics of selected immune cells responsible for ADA formation. It also successfully captured the impact of tacrolimus/sirolimus immunomodulation on ADA formation, demonstrating that the regimen effectively suppressed ADA formations and induced immune tolerance. Conclusions: This work demonstrates the utility of modeling approaches to integrate pharmacokinetic and immunogenicity assessment data for the prospective planning of long-term toxicology studies to support the preclinical development of mAbs.
Keywords: anti-drug antibodies; immune tolerance; monoclonal antibodies; pharmacokinetics; population pharmacokinetic model; preclinical; semi-mechanistic model.
Conflict of interest statement
P.G. and B.M. declare no conflicts of interest. J.R. is an employee of EMD Serono and owns EMD Serono stock. V.J. is an employee of Bristol Myers Squibb and owns Bristol Myers Squibb stock.
Figures


Similar articles
-
Short-Term Immunosuppression in Rats Induces Prolonged Immune Tolerance Towards a Human Monoclonal Antibody, Erenumab.AAPS J. 2025 May 16;27(4):94. doi: 10.1208/s12248-025-01083-0. AAPS J. 2025. PMID: 40379907
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2020 Jan 9;1(1):CD011535. doi: 10.1002/14651858.CD011535.pub3. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Apr 19;4:CD011535. doi: 10.1002/14651858.CD011535.pub4. PMID: 31917873 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Clinical and cost-effectiveness of newer immunosuppressive regimens in renal transplantation: a systematic review and modelling study.Health Technol Assess. 2005 May;9(21):1-179, iii-iv. doi: 10.3310/hta9210. Health Technol Assess. 2005. PMID: 15899149
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
References
-
- Crommelin D.J.A., Sinderlar R.D., Meibohm B. Pharmaceutical Biotechnology: Fundamentals and Applications. 6th ed. Springer Nature; Cham, Switzerland: 2024.
LinkOut - more resources
Full Text Sources
Research Materials

