Source of Explant and Light Spectrum Influence in Adventitious Shoot Regeneration of Prunus salicina Lindl. (Japanese plum)
- PMID: 40733467
- PMCID: PMC12298175
- DOI: 10.3390/plants14142230
Source of Explant and Light Spectrum Influence in Adventitious Shoot Regeneration of Prunus salicina Lindl. (Japanese plum)
Abstract
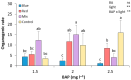
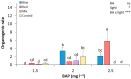
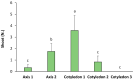
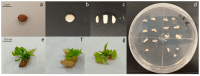
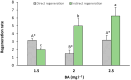
Light influence on shoot regeneration in Prunus salicina is a complex interaction that has been studied for the first time. Japanese plum plants were regenerated from calli and seeds of the scion cultivar 'Victoria'. The effect of four different light spectra (white, blue, red, and mixed), along with three 6-benzyladenine (BA) concentrations (1, 1.5, and 2 mg L-1), was studied in these two sources of explants. Organogenic calli were derived from the base of stem explants of the scion cultivar 'Victoria', whereas cotyledons and embryogenic axis slices were used as seed explants. Calli cultured with 2 mg L-1 of BA and mixed light or 2.5 mg L-1 of BA and control light showed the highest regeneration rates, with no significant differences compared to other treatments. Seed explants exposed to 2.5 mg L-1 of BA and red light exhibited significantly higher organogenesis. In comparison, those in 1.5 mg L-1 of BA with blue light or 2.5 mg L-1 of BA with mixed/control light showed no regeneration. BA concentration did not have a significant effect in the induction of somatic shoots from any explant source. In contrast, a strong interaction between light and BA was noticed. This work presents a protocol that can be applied in transformation and editing research as light spectrum studies continue to advance.
Keywords: 6-benzyladenine; LEDs; de novo regeneration; morphogenesis; somatic shoots.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Canli F.A., Tian L. Regeneration of adventitious shoots from mature stored cotyledons of Japanese plum (Prunus salicina Lindl.) Sci. Hortic. 2009;120:64–69. doi: 10.1016/j.scienta.2008.09.017. - DOI
-
- Pérez-Clemente R.M., Pérez-Sanjuán A., García-Férriz L., Beltrán J.P., Cañas L.A. Transgenic peach plants (Prunus persica L.) produced by genetic transformation of embryo sections using the green fluorescent protein (GFP) as an in vivo marker. Mol. Breed. 2005;14:419–427. doi: 10.1007/s11032-005-0506-5. - DOI
-
- Tian L., Wen Y., Jayasankar S., Sibbald S. Regeneration of Prunus salicina Lindl. (Japanese plum) from hypocotyls of mature seeds. Vitr. Cell. Dev. Biol. Plant. 2007;43:343–347. doi: 10.1007/s11627-007-9047-4. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

