Adverse Events and Associated Economic Burden of COVID-19 Vaccination in Queensland, Australia: Findings from the Cross-Sectional QoVAX-Statewide Study
- PMID: 40733689
- PMCID: PMC12300968
- DOI: 10.3390/vaccines13070712
Adverse Events and Associated Economic Burden of COVID-19 Vaccination in Queensland, Australia: Findings from the Cross-Sectional QoVAX-Statewide Study
Abstract
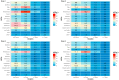
Background/Objectives: The economic impact of adverse events following COVID-19 immunisation (AEFIs) in Australia is underexplored. This study aimed to assess the economic burden of AEFIs on both healthcare systems and societal productivity. Methods: A cross-sectional survey was conducted in Queensland residents aged ≥18 years who had received at least one dose of a COVID-19 vaccine in the preceding 12 months. Overall, 6964 participants were recruited from July to September 2022 via email and broad social media campaigns. The survey collected data on the incidence, type and duration of AEFIs; healthcare utilisation; and work-related absenteeism. Healthcare costs were estimated using national healthcare reimbursement data, and productivity costs were estimated using Australian Bureau of Statistics Average Weekly Earnings. Results: Of the 6797 eligible respondents (predominantly female [62%]; median age: 52 years), AEFIs were reported by 53.4%, 44.1%, 40.7%, and 40.9% following doses 1 to 4, respectively. Pain and tenderness were predominant local AEFIs, while tiredness and headaches were the most frequent systemic AEFIs, generally resolving within three days. Relatively few participants reporting AEFIs consulted medical professionals: 7.0%, 7.3%, 5.1%, and 1.9% following each dose, respectively. The mean healthcare cost per person reporting AEFIs was AUD 24, AUD 88, AUD 22, and AUD 4 following each respective dose. Work absenteeism was recorded in 16.5%, 18.2%, 15.2%, and 11.2% following each dose with mean absenteeism days per person of 4.7, 7.4, 3.6 and 2.1, respectively, and mean productivity costs per person reporting AEFIs amounting to AUD 1494, AUD 2388, AUD 1136, and AUD 690, respectively. Conclusions: Participants reported mostly mild AEFIs with only a small proportion of individuals seeking medical services. Productivity costs attributable to these AEFIs exceeded direct healthcare expenses incurred.
Keywords: COVID-19; adverse events; economic analysis; productivity loss; vaccination.
Conflict of interest statement
The authors declare no actual of perceived conflict of interest relevant to this research.
Figures
Similar articles
-
Delivery of intravenous anti-cancer therapy at home versus in hospital or community settings for adults with cancer.Cochrane Database Syst Rev. 2025 Apr 22;4(4):CD014861. doi: 10.1002/14651858.CD014861.pub2. Cochrane Database Syst Rev. 2025. PMID: 40260850
-
Measures implemented in the school setting to contain the COVID-19 pandemic.Cochrane Database Syst Rev. 2022 Jan 17;1(1):CD015029. doi: 10.1002/14651858.CD015029. Cochrane Database Syst Rev. 2022. Update in: Cochrane Database Syst Rev. 2024 May 2;5:CD015029. doi: 10.1002/14651858.CD015029.pub2. PMID: 35037252 Free PMC article. Updated.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
References
-
- Australian National Audit Office (ANAO) Australia’s COVID-19 Vaccine Rollout. [(accessed on 6 June 2024)]; Available online: https://www.anao.gov.au/work/performance-audit/australia-covid-19-vaccin....
LinkOut - more resources
Full Text Sources

