Research on comprehensive quality consistency evaluation strategy for TCM Granules: A case study with sugar-free Yangwei Granules produced by fluid-bed granulation
- PMID: 40734905
- PMCID: PMC12301915
- DOI: 10.1016/j.chmed.2024.11.001
Research on comprehensive quality consistency evaluation strategy for TCM Granules: A case study with sugar-free Yangwei Granules produced by fluid-bed granulation
Abstract
Objective: To develop a quality consistency evaluation strategy for traditional Chinese medicine (TCM) granules using sugar free Yangwei Granules as a model drug, and demonstrate the effectiveness of the developed method.
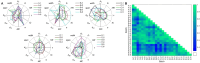
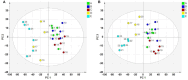
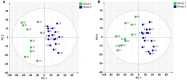
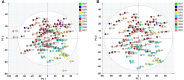
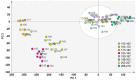
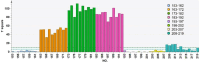
Methods: The strategy integrates several methods including, HPLC fingerprint and physical fingerprint methods analyze the similarity in chemical and physical properties of the TCM granule samples. Near-infrared (NIR) spectroscopy with principal components cluster analysis method is used to monitor normal operating conditions (NOC) samples accurately and to identify different types of abnormal operating conditions (AOC) samples, particularly those that deviate from the normal range.
Results: The combined use of HPLC fingerprint and physical fingerprint provides insights into the chemical and physical properties of the samples. NIR spectroscopy, combined with principal components cluster analysis, achieves high accuracy in monitoring NOC samples and identifying AOC samples without misjudgment. The approach proves useful as a complementary method in cases where HPLC fingerprint and physical fingerprint alone lack sufficient resolution.
Conclusion: This study establishes the feasibility and utility of the integrated approach for assessing the quality consistency of TCM granules. The strategy shows a high degree of generalization and holds significant importance for enhancing the quality control processes of TCM granules.
Keywords: chemometrics; multivariate analysis; near-infrared spectroscopy; quality consistency evaluation strategy; sugar-free Yangwei Granules; traditional Chinese medicine granules.
© 2024 Tianjin Press of Chinese Herbal Medicines. Published by ELSEVIER B.V.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

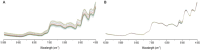


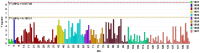
Similar articles
-
Management of urinary stones by experts in stone disease (ESD 2025).Arch Ital Urol Androl. 2025 Jun 30;97(2):14085. doi: 10.4081/aiua.2025.14085. Epub 2025 Jun 30. Arch Ital Urol Androl. 2025. PMID: 40583613 Review.
-
The effectiveness and cost-effectiveness of carmustine implants and temozolomide for the treatment of newly diagnosed high-grade glioma: a systematic review and economic evaluation.Health Technol Assess. 2007 Nov;11(45):iii-iv, ix-221. doi: 10.3310/hta11450. Health Technol Assess. 2007. PMID: 17999840
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Chinese herbal medicine for oesophageal cancer.Cochrane Database Syst Rev. 2016 Jan 22;2016(1):CD004520. doi: 10.1002/14651858.CD004520.pub7. Cochrane Database Syst Rev. 2016. PMID: 26799001 Free PMC article.
-
Evaluation of impact on health-related quality of life and cost effectiveness of Traditional Chinese Medicine: a systematic review of randomized clinical trials.J Altern Complement Med. 2012 Dec;18(12):1108-20. doi: 10.1089/acm.2011.0315. Epub 2012 Aug 27. J Altern Complement Med. 2012. PMID: 22924383
References
-
- Fransson M., Folestad S. Real-time alignment of batch process data using COW for on-line process monitoring. Chemometrics & Intelligent Laboratory Systems. 2006;84(1):56–61.
-
- Li L.F., Zhang Q.W., Han Q.B. Recent advances in qualitative and quantitative analysis of polysaccharides in natural medicines: A critical review. Journal of Pharmaceutical and Biomedical Analysis. 2022;220 - PubMed
-
- Li X., Yang H., Pang X., Sun G. Entirely control the quality consistency of Rong'e Yishen oral liquid by both quantified profiling and quantitative analysis of multi-components by single marker method. Journal of Pharmaceutical Biomedical Analysis. 2021;193 - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

