Relationship between brain atrophy and disability in a multi-site multiple sclerosis registry
- PMID: 40734995
- PMCID: PMC12306358
- DOI: 10.1136/bmjno-2025-001126
Relationship between brain atrophy and disability in a multi-site multiple sclerosis registry
Abstract
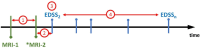
Background: In a retrospective multicentre cohort study, we explored the association between brain atrophy and multiple sclerosis (MS) disability using different MRI scanners and protocols at multiple sites.
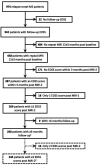
Methods: Relapse-onset MS patients were included if they had two clinical MRIs 12 months apart and ≥2 Expanded Disability Status Scale (EDSS) scores. Percentage brain volume change (PBVC), percentage grey matter change (PGMC), fluid-attenuated inversion recovery (FLAIR) lesion volume change, whole brain volume (BV), grey matter volume (GMV), FLAIR lesion volume and T1 hypointense lesion volume were assessed by icobrain. Disability was measured by EDSS scores and 6-month confirmed disability progression (CDP).
Results: Of the 260 relapse-onset MS patients included, 204 (78%) MRI pairs were performed in the same scanner and 56 (22%) pairs were from different scanners. 93% of patients were on treatment and mean PBVC was -0.26% (±0.52). During the median follow-up of 2.8 years from the second MRI, median EDSS change was 0.0 and 12% patients experienced 6-month CDP. Cross-sectional BV and GMV at the later MRI showed a trend for association with CDP (HR 0.99; 95% CI 0.98 to 1.00; p=0.06). Only BV at the later MRI was associated with EDSS score (β -0.03, SE 0.01, p<0.001) and the rate of EDSS change over time (β -0.001, SE 0.0003, p=0.02). There was no association between longitudinal PBVC or PGMC and CDP or EDSS (p>0.05).
Conclusion: In this highly treated MS cohort with low disability accrual, only cross-sectional BV showed an association with future EDSS scores, while no MRI metric predicted 6-month CDP. These findings highlight the limitations of current clinical MRI measures in predicting disability worsening in real-world settings.
Keywords: MRI; MULTIPLE SCLEROSIS.
Copyright © Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
A-LN received research grants from Novartis, Biogen, Merck Serono and MS Research Australia; speaker honoraria and consulting fees from Biogen, Teva and Merck Serono; conference travel support from Genzyme-Sanofi, Biogen and Roche. DH was supported by the Czech Ministry of Education project Progres Q27/LF1. She also received compensation for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck, Bayer, Sanofi Genzyme, Roche and Teva, as well as support for research activities from Biogen Idec. EHH received honoraria/research support from Biogen, Merck Serono, Novartis, Roche and Teva; served on advisory boards for Actelion, Biogen, Celgene, Merck Serono, Novartis and Sanofi Genzyme; has been supported by the Czech Ministry of Education research project PROGRES Q27/LF1. MBar has received institutional support for research, speaking and/or participation in advisory boards (Biogen, Novartis and Sanofi Genzyme); and is a research consultant (RxMx and Sydney Neuroimaging Analysis Centre). MPS received consulting fees from Biogen, Sanofi Genzyme, Roche, Novartis, Merck, GeNeuro, Celgene, TEVA and Medday. NDS has received honoraria from Biogen-Idec, Celgene, Immunic, Merck Serono, Novartis, Roche, Sanofi-Genzyme and Teva for consulting services, speaking and travel support. He serves on advisory boards for Biogen-Idec, Immunic, Merck Serono, Novartis, Roche and Sanofi-Genzyme. He has received research grant support from the Italian Multiple Sclerosis Society. MBat: none. MV was supported by the Czech Ministry of Health project MH CZ-DRO-VFN64165. She also received compensation for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Sanofi Genzyme, Roche and Teva, as well as support for research activities from Biogen Idec. EL: none. FG is the founder and CEO of Radiopaedia Australia Pty Ltd. Speaker fees from Sanofi-Genzyme. Grant funding from Royal Melbourne Hospital Foundation, Royal Australian and New Zealand College of Radiology. PMD: none. HP: none. MD: none. AvdW has received travel support and served on advisory boards for Novartis, Biogen, Merck Serono, Roche and Teva. She receives grant support from the National Health and Medical Research Council of Australia. VGJ received conference travel support from Roche and Merck; and speaker honoraria from Roche and Biogen. She receives research grant support from MS Research Australia and the National Health and Medical Research Council of Australia (NHMRC 1156519). FPo is an employee of icometrix. RZ received personal compensation from EMD Serono, Sanofi, Bristol Myers Squibb, Keystone Heart and Novartis for speaking and consulting fees. He received financial support for research activities from Novartis, Mapi Pharma, Bristol Myers Squibb, Protembis, V-Wave Medical and Keystone Heart. BW-G has participated in speaker’s bureaus and/or served as a consultant for Biogen, EMD Serono, Novartis, Genentech, Celgene, Abbvie, Sanofi and Mallinckrodt. BW-G also has received grant/research support from the agencies listed in the previous sentence. She serves on the editorial board for BMJ Neurology, Frontiers in Epidemiology, Journal of MS and CNS Drugs. MBD’h has served on medical advisory boards for Roche, Sanofi-Genzyme, Biogen, Merck, Bayer-Schering and Novartis. She received restricted grants and/or congress support from Biogen, Merck, Teva, Roche, Novartis and Bayer-Schering. GN is medical director neurology at icometrix. He or his institution (VUB/UZ Brussel) has received research, educational and travel grants from Biogen, Roche, Genzyme, Merck, Bayer and Teva. VVP has received travel grants from Merck, Biogen, Sanofi, Celgene, Almirall and Roche. His institution has received research grants and consultancy fees from Roche, Biogen, Sanofi, Celgene, Merck and Novartis Pharma. GL: none. LVH: none. JL-S has received travel compensation from Biogen, Merck, Novartis; has been involved in clinical trials with Biogen, Novartis, Roche. Her institution has received honoraria for talks and advisory board service from Biogen, Merck, Novartis. FPa has received compensation for consulting, talks, advisory/steering board activities from Almirall, Bayer, Biogen, Merck, Novartis, Sanofi-Genzyme and TEVA; research support from Biogen, Merck, University of Catania, RELOAD Onlus and FISM. EC received compensation for travel, speaker honoraria, consultant fees as well as support for research activities from Biogen Idec, Novartis, Merck, Bayer, Sanofi Genzyme, Roche and Teva. JIR has received fees in concept of honoraria speaker and grant for research from Biogen, Sanofi-Genzyme, Novartis, Merck, Bayer and Roche. DMS is an employee of icometrix. WVH is CEO, founder, shareholder and member of the board of icometrix. WVH is CEO, founder, shareholder and member of the board of icometrix. TK served on scientific advisory boards or as a consultant for MS International Federation and WHO, Therapeutic Goods Administration, BMS, Roche, Janssen, Genzyme, Novartis, Merck and Biogen, received conference travel support and/or speaker honoraria from WebMD Global, Merck, Sandoz, Novartis, Biogen, Roche, Eisai, Genzyme, Teva and BioCSL and received research or educational event support from Biogen, Novartis, Genzyme, Roche, Celgene and Merck. HB’s institution receives compensation for Advisory Board, Steering Committee and Educational activities from Biogen, Roche, Merck and Novartis. His institution receives research support from Roche, Novartis, Biogen, NHMRC and MRFF Australia, MS Research Australia. He receives personal compensation from Oxford HPF for serving on the steering group of MS Brain Health.
Figures
References
LinkOut - more resources
Full Text Sources
