LARGE protein drives activity-induced homeostatic resetting
- PMID: 40737424
- PMCID: PMC12309686
- DOI: 10.1126/sciadv.adt0703
LARGE protein drives activity-induced homeostatic resetting
Abstract
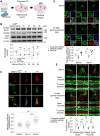
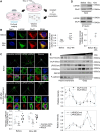
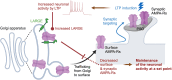
In the brain, memory can be coded as relative differences in synaptic strength produced by Hebbian plasticity [e.g., long-term potentiation (LTP)]. However, changes in neuronal activity, including the saturation of synaptic strength by the positive-feedback nature of Hebbian plasticity, could deteriorate the encoded memory. Homeostatic plasticity is thought to contribute to the stability of the encoded memory by maintaining the relative differences in synaptic strength against persistent destabilizing changes in neuronal activity. However, it remains unclear how and when these two types of plasticity work together in the context of memory. Here, we have demonstrated that LARGE, a protein associated with intellectual disability, drives homeostatic resetting several hours after LTP by down-regulating AMPA-receptor trafficking via the Golgi apparatus. LARGE deficiency impairs long-term memory formation in mice. Our study reveals a potential molecular mechanism underlying the stability of memory mediated by cross-talk between Hebbian and homeostatic plasticity.
Figures





References
-
- Boudkkazi S., Schwenk J., Nakaya N., Brechet A., Kollewe A., Harada H., Bildl W., Kulik A., Dong L., Sultana A., Zolles G., Schulte U., Tomarev S., Fakler B., A Noelin-organized extracellular network of proteins required for constitutive and context-dependent anchoring of AMPA-receptors. Neuron 111, 2544–2556.e9 (2023). - PMC - PubMed
-
- Hanley J. G., Endosomal sorting of AMPA receptors in hippocampal neurons. Biochem. Soc. Trans. 38, 460–465 (2010). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

