Machine Learning Model for Predicting Pathological Invasiveness of Pulmonary Ground-Glass Nodules Based on AI-Extracted Radiomic Features
- PMID: 40745923
- PMCID: PMC12313823
- DOI: 10.1111/1759-7714.70128
Machine Learning Model for Predicting Pathological Invasiveness of Pulmonary Ground-Glass Nodules Based on AI-Extracted Radiomic Features
Abstract
Background: With the widespread adoption of low-dose CT screening, the detection of pulmonary ground-glass nodules (GGNs) has risen markedly, presenting diagnostic challenges in distinguishing preinvasive lesions from invasive adenocarcinomas (IAC). This study aimed to develop a machine learning (ML)-based model using artificial intelligence (AI)-extracted CT radiomic features to predict the invasiveness of GGNs.
Methods: A retrospective cohort of 285 patients (148 with preinvasive lesions, 137 with IAC) from the Lingnan Campus was divided into training and internal validation sets (8:2). An independent cohort of 210 patients (118 with preinvasive lesions, 92 with IAC) from the Tianhe Campus served as external validation. Nineteen radiomic features were extracted and filtered using Boruta and LASSO algorithms. Seven ML classifiers were evaluated using AUC-ROC, decision curve analysis (DCA), and SHAP interpretability.
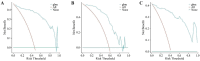
Results: Median CT value, skewness, 3D long-axis diameter, and transverse diameter were ultimately selected for model construction. Among all classifiers, the Gradient Boosting Machine (GBM) model achieved the best performance (AUC = 0.965 training, 0.908 internal validation, and 0.965 external validation). It demonstrated strong accuracy (88.1%), specificity (80.7%), and F1 score (0.87) in the external validation cohort. The GBM model demonstrated superior net clinical benefit. SHAP analysis identified median CT value and skewness as the most influential predictors.
Conclusion: This study presents a simplified ML model using AI-extracted radiomic features, which has strong predictive performance and biological interpretability for preoperative risk stratification of GGNs. By leveraging median CT value, skewness, 3D long-axis diameter, and transverse diameter, the model enables accurate and noninvasive differentiation between IAC and indolent lesions, supporting precise surgical planning.
Keywords: artificial intelligence; invasiveness; pulmonary ground‐glass nodules; radiomics.
© 2025 The Author(s). Thoracic Cancer published by John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
The Value of Topological Radiomics Analysis in Predicting Malignant Risk of Pulmonary Ground-Glass Nodules: A Multi-Center Study.Technol Cancer Res Treat. 2024 Jan-Dec;23:15330338241287089. doi: 10.1177/15330338241287089. Technol Cancer Res Treat. 2024. PMID: 39363876 Free PMC article.
-
A Predictive Model Integrating AI Recognition Technology and Biomarkers for Lung Nodule Assessment.Thorac Cardiovasc Surg. 2025 Mar;73(2):174-181. doi: 10.1055/a-2446-9832. Epub 2024 Nov 26. Thorac Cardiovasc Surg. 2025. PMID: 39591993 Free PMC article.
-
Ensemble Machine Learning Classifiers Combining CT Radiomics and Clinical-Radiological Features for Preoperative Prediction of Pathological Invasiveness in Lung Adenocarcinoma Presenting as Part-Solid Nodules: A Multicenter Retrospective Study.Technol Cancer Res Treat. 2025 Jan-Dec;24:15330338251351365. doi: 10.1177/15330338251351365. Epub 2025 Jun 17. Technol Cancer Res Treat. 2025. PMID: 40525253 Free PMC article.
-
The Machine Learning Models in Major Cardiovascular Adverse Events Prediction Based on Coronary Computed Tomography Angiography: Systematic Review.J Med Internet Res. 2025 Jun 13;27:e68872. doi: 10.2196/68872. J Med Internet Res. 2025. PMID: 40513092 Free PMC article.
-
Progress in the application of machine learning in CT diagnosis of acute appendicitis.Abdom Radiol (NY). 2025 Sep;50(9):4040-4049. doi: 10.1007/s00261-025-04864-5. Epub 2025 Mar 17. Abdom Radiol (NY). 2025. PMID: 40095017 Review.
References
-
- Liu Z., Liu X., and Ni L., “Analysis of Pulmonary Nodules Detected by Annual Low‐Dose Computed Tomography in the Elderly During a 10‐Year Follow‐Up,” Geriatrics & Gerontology International 22, no. 10 (2022): 865–869. - PubMed
-
- Gould M. K., Tang T., Liu I. L., et al., “Recent Trends in the Identification of Incidental Pulmonary Nodules,” American Journal of Respiratory and Critical Care Medicine 192, no. 10 (2015): 1208–1214. - PubMed
-
- Ye T., Deng L., Wang S., et al., “Lung Adenocarcinomas Manifesting as Radiological Part‐Solid Nodules Define a Special Clinical Subtype,” Journal of Thoracic Oncology 14, no. 4 (2019): 617–627. - PubMed
-
- Tsao M. S., Nicholson A. G., Maleszewski J. J., Marx A., and Travis W. D., “Introduction to 2021 WHO Classification of Thoracic Tumors,” Journal of Thoracic Oncology 17, no. 1 (2022): e1–e4. - PubMed
-
- Travis W. D., Brambilla E., Nicholson A. G., et al., “The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification,” Journal of Thoracic Oncology 10, no. 9 (2015): 1243–1260. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

