Simultaneous remediation of As(V), Cd(II) and Pb(II) in aqueous solution and soil using Fe/Ca-modified biochar
- PMID: 40746064
- PMCID: PMC12301511
- DOI: 10.1177/0734242X241307521
Simultaneous remediation of As(V), Cd(II) and Pb(II) in aqueous solution and soil using Fe/Ca-modified biochar
Abstract
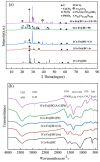
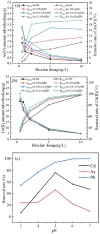
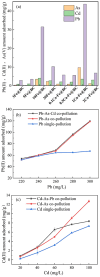
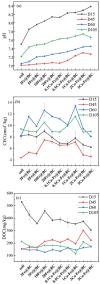
A novel Fe/Ca-modified biochar (BC) derived from corn stalk was prepared in this study for the simultaneous adsorption and immobilization of arsenic, cadmium and lead in aqueous solution and soil, respectively. The adsorption experiment in aqueous solution indicated that 1Ca-Fe@BC exhibited excellent removal efficiency (R, %) towards arsenic (46.3%), cadmium (76.3%) and lead (93.6%). The presence of cadmium and lead could enhance the adsorption efficiency of arsenic by facilitating the formation of ternary complexes with it. Moreover, both cooperative and competitive effects occurred between cadmium and lead adsorption. When the distribution coefficient of cadmium and lead was equimolar, cooperative adsorption prevailed. However, when the distribution coefficient deviated from this balance, competitive adsorption dominated with a higher affinity for lead. The soil culture experiment demonstrated that the utilization of 1Ca-Fe@BC could enhance the stability of arsenic, cadmium and lead in soil by enhancing soil cation exchange capacity and pH while reducing dissolved organic carbon. The competitive and cooperative effects of arsenic, cadmium and lead in soil were consistent with those found in aqueous solution. The results showed that 1Ca-Fe@BC exhibited an excellent remediation efficiency for both arsenic, cadmium and lead-contaminated aqueous solution and soil.
Keywords: Fe/Ca modified; Multi-contamination; biochar; heavy metal; simultaneous remediation.
Conflict of interest statement
Declaration of conflicting interestsThe authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures


Similar articles
-
Iron-silicon modified biochar for remediation of cadmium/arsenic co-contaminated paddy fields: Is it possible to kill two birds with one stone?J Hazard Mater. 2025 Aug 15;494:138702. doi: 10.1016/j.jhazmat.2025.138702. Epub 2025 May 21. J Hazard Mater. 2025. PMID: 40435623
-
Impact of citric acid on cadmium immobilization in soil amended with biochar.J Environ Sci (China). 2025 Dec;158:324-336. doi: 10.1016/j.jes.2025.03.005. Epub 2025 Mar 8. J Environ Sci (China). 2025. PMID: 40841014
-
Revitalizing cadmium-stressed sunflower: co-composted biochar improves growth, antioxidant responses, and soil remediation efficiency.BMC Plant Biol. 2025 Jul 4;25(1):875. doi: 10.1186/s12870-025-06906-y. BMC Plant Biol. 2025. PMID: 40615980 Free PMC article.
-
AI-assisted systematic review on remediation of contaminated soils with PAHs and heavy metals.J Hazard Mater. 2024 Apr 15;468:133813. doi: 10.1016/j.jhazmat.2024.133813. Epub 2024 Feb 17. J Hazard Mater. 2024. PMID: 38402679
-
Removal of vapor-phase elemental mercury from stack emissions with sulfur-impregnated activated carbon.Rev Environ Contam Toxicol. 2014;230:1-34. doi: 10.1007/978-3-319-04411-8_1. Rev Environ Contam Toxicol. 2014. PMID: 24609516
References
-
- Amen R, Bashir H, Bibi I, et al. (2020) A critical review on arsenic removal from water using biochar-based sorbents: The significance of modification and redox reactions. Chemical Engineering Journal 396: 125195.
-
- Arancibia-Miranda N, Manquian-Cerda K, Pizarro C, et al. (2020) Mechanistic insights into simultaneous removal of copper, cadmium and arsenic from water by iron oxide-functionalized magnetic imogolite nanocomposites. Journal of Hazardous Materials 398: 122940. - PubMed
-
- Arcibar-Orozco JA, Rangel-Mendez JR, Diaz-Flores PE. (2015) Simultaneous adsorption of Pb(II)-Cd(II), Pb(II)-phenol, and Cd(II)-phenol by activated carbon cloth in aqueous solution. Water Air and Soil Pollution 226: 2197.
-
- Beesley L, Marmiroli M. (2011) The immobilisation and retention of soluble arsenic, cadmium and zinc by biochar. Environmental Pollution 159: 474–480. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

