Efficacy of third-line chemotherapy following nanoliposomal irinotecan combined with fluorouracil and folinic acid as second-line treatment for unresectable pancreatic cancer
- PMID: 40746600
- PMCID: PMC12310504
- DOI: 10.3389/fonc.2025.1626689
Efficacy of third-line chemotherapy following nanoliposomal irinotecan combined with fluorouracil and folinic acid as second-line treatment for unresectable pancreatic cancer
Abstract
Introduction: The significance of third-line chemotherapy (CTx) in unresectable pancreatic cancer (UPC) remains unclear. This study evaluated the therapeutic impact of third-line CTx after nanoliposomal irinotecan and fluorouracil combined with folinic acid (nal-IRI + 5-FU/LV) therapy as second-line CTx for UPC.
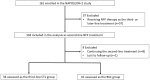
Methods: Between June 2020 and May 2021, 104 patients who received nal-IRI + 5-FU/LV therapy as second-line CTx were retrospectively analyzed for post-discontinuation survival (PDS) and overall survival (OS). Comparisons were made between patients transitioning to third-line CTx and those receiving best supportive care (BSC), using a Cox proportional hazards model adjusted for patient background.
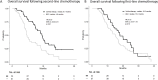
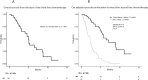
Results: Of the cohort, 34 patients received third-line CTx, whereas 61 transitioned to BSC. The median OS from first-line CTx in the third-line CTx group was 18.0 months, with a median OS of 9.7 months from second-line CTx. Adjusted median PDS following second-line CTx was 6.5 months for the third-line CTx group compared to 2.3 months for the BSC group (adjusted hazard ratio 0.16; 95% confidence interval 0.08-0.32; P < 0.01).
Conclusion: Third-line CTx should be actively considered for patients with UPC, as the approach may significantly extend survival in those who can tolerate the treatment.
Keywords: best supportive care; chemotherapy; nanoliposomal irinotecan; pancreatic cancer; third-line treatment.
Copyright © 2025 Miwa, Kawasaki, Shimokawa, Otsuka, Tanaka, Fukahori, Shibuki, Nakazawa, Arima, Koga, Ueda, Kubotsu, Shimokawa, Takeshita, Nishikawa, Komori, Otsu, Hosokawa, Sakai, Oda, Kawahira, Arita, Honda, Taguchi, Tsuneyoshi, Fujita, Sakae, Kawaguchi, Shirakawa, Mizuta and Mitsugi.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Hori M, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H, et al. Cancer incidence and incidence rates in Japan in 2009: A study of 32 population-based cancer registries for the monitoring of cancer incidence in Japan (Mcij) project. Jpn J Clin Oncol. (2015) 45:884–91. doi: 10.1093/jjco/hyv088, PMID: - DOI - PubMed
-
- Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: A phase iii trial of the national cancer institute of Canada clinical trials group. J Clin Oncol. (2007) 25:1960–6. doi: 10.1200/JCO.2006.07.9525, PMID: - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

