Pentosan polysulfate alleviates interstitial cystitis/bladder pain syndrome by modulating bile acid metabolism and activating the TGR5 receptor through gut microbiota regulation
- PMID: 40747461
- PMCID: PMC12308114
- DOI: 10.14440/bladder.2024.0060
Pentosan polysulfate alleviates interstitial cystitis/bladder pain syndrome by modulating bile acid metabolism and activating the TGR5 receptor through gut microbiota regulation
Abstract
Background: The disrupted gut microbiome has been found to be implicated in the development of interstitial cystitis/bladder pain syndrome (IC/BPS). Pentosan polysulfate (PPS) is an oral medication used for treating IC/BPS, acting as both an anti-inflammatory agent and a bladder barrier protector. However, the precise mechanisms by which the PPS-mediated modulation of the gut microbiome alleviates IC/BPS are not fully understood.
Objective: This study aimed to identify the key gut microbiota species and metabolites involved in PPS's protective effects against IC/BPS.
Methods: We employed a multifaceted approach, including 16S rDNA gene sequencing, antibiotic treatment, and fecal microbiota transplantation, to validate the dependency of PPS's protective effects on the gut microbiome. Furthermore, we performed a comprehensive metabolomic profiling using non-targeted metabolomics and liquid chromatography-tandem mass spectrometry.
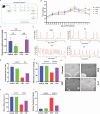
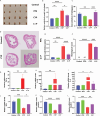
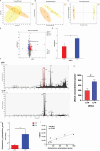
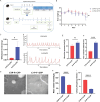
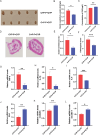
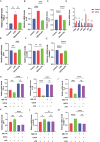
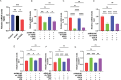
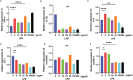
Results: PPS significantly elevated the abundance of the xylan-degrading bacteria, Eubacterium xylanophilum group, which, through its interaction with the gut microbiome, markedly reduced inflammation and barrier damage induced by cyclophosphamide in IC/BPS. In addition, PPS significantly increased the level of ursodeoxycholic acid (UDCA), a secondary bile acid, demonstrating a strong correlation with the abundance of the E. xylanophilum group. Ex vivo supplementation with UDCA mitigated lipopolysaccharide-induced inflammation and barrier disruption in SV-HUC-1 cells by activating the TGR5 receptor.
Conclusion: PPS exerts its protective effects against IC/BPS by modulating the gut microbiome and its metabolites.
Keywords: Bladder barrier; Cyclophosphamide; Gut microbiota; Interstitial cystitis/bladder pain syndrome; Pentosan polysulfate; Ursodeoxycholic acid.
Copyright: © 2025 Author(s).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
[Pentosan polysulfate alleviates cyclophosphamide-induced interstitial cystitis/bladder pain syndrome in mice by modulating gut microbiota and bile acid metabolism].Nan Fang Yi Ke Da Xue Xue Bao. 2025 Jun 20;45(6):1270-1279. doi: 10.12122/j.issn.1673-4254.2025.06.16. Nan Fang Yi Ke Da Xue Xue Bao. 2025. PMID: 40579140 Free PMC article. Chinese.
-
Interventions for treating people with symptoms of bladder pain syndrome: a network meta-analysis.Cochrane Database Syst Rev. 2020 Jul 30;7(7):CD013325. doi: 10.1002/14651858.CD013325.pub2. Cochrane Database Syst Rev. 2020. PMID: 32734597 Free PMC article.
-
The microbiota in patients with interstitial cystitis/bladder pain syndrome: a systematic review.BJU Int. 2024 Dec;134(6):869-880. doi: 10.1111/bju.16439. Epub 2024 Jun 18. BJU Int. 2024. PMID: 38890150
-
Oxidative Stress and Chronic Inflammation as Partners in Crime in Interstitial Cystitis/Bladder Pain Syndrome.J Innate Immun. 2025;17(1):369-396. doi: 10.1159/000546901. Epub 2025 Jun 26. J Innate Immun. 2025. PMID: 40570823 Free PMC article. Review.
-
Dietary curcumin supplementation enhances growth performance and anti-inflammatory functions by modulating gut microbiota, microbiota-derived metabolites, and expression of inflammation-related genes in broilers.J Anim Sci. 2024 Jan 3;102:skae296. doi: 10.1093/jas/skae296. J Anim Sci. 2024. PMID: 39324614
References
-
- Clemens JQ, Erickson DR, Varela NP, Lai HH.Diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol 2022;208(1):34–42. doi: 10.1097/JU.0000000000002756 - PubMed
-
- Di XP, Peng L, Xiang LY, Wang MH, Zhang J, Luo DY.A bibliometric analysis of top-cited journal articles in interstitial cystitis and bladder pain syndrome. Int Urogynecol J 2022;33(9):2557–2563. doi: 10.1007/s00192-022-05298-z - PubMed
-
- Mohammad A, Laboulaye MA, Shenhar C, Dobberfuhl AD.Mechanisms of oxidative stress in interstitial cystitis/bladder pain syndrome. Nat Rev Urol 2024;21(7):433–449. doi: 10.1038/s41585-023-00850-y - PubMed
-
- Kayama H, Okumura R, Takeda K.Interaction between the microbiota, epithelia, and immune cells in the intestine. Annu Rev Immunol 2020;38:23–48. doi: 10.1146/annurev-immunol-070119-115104 - PubMed
LinkOut - more resources
Full Text Sources
