Development of a Vaginal Extracellular Matrix Hydrogel for Combating Genitourinary Syndrome of Menopause
- PMID: 40751544
- PMCID: PMC12463450
- DOI: 10.1002/adma.202419977
Development of a Vaginal Extracellular Matrix Hydrogel for Combating Genitourinary Syndrome of Menopause
Abstract
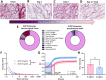
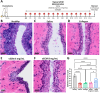
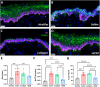
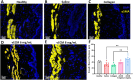
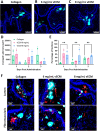
Genitourinary syndrome of menopause (GSM) is initiated by the hypoestrogenic state consequent to cessation of ovarian function and involves changes in all vaginal layers. These alterations include decreased epithelial proliferation and differentiation, impaired extracellular matrix (ECM) maintenance, pro-inflammatory immune milieu, and smooth muscle atrophy. Patient satisfaction with existing treatments for GSM is low, due in part to their lack of therapeutic effect throughout all vaginal layers and reliance on hormonal treatments. Decellularized ECM therapeutics are known to facilitate soft tissue repair in a variety of applications. Thus, a porcine vaginal tissue-derived decellularized ECM (vECM) hydrogel suitable for noninvasive topical intravaginal administration is developed specifically for vaginal repair. When applied in a rat model of surgical menopause, vECM significantly improves vaginal epithelial thickness and epithelial stem cell phenotype. Despite topical intravaginal application, vECM is also observed in the lamina propria and fibromuscularis, exerting a regenerative effect on the vaginal smooth muscle layer. Furthermore, vECM modulates macrophage density and promotes pro-regenerative M2-like phenotype within the vaginal tissue. Overall, the work herein demonstrates a new nonhormonal biomaterial treatment that counteracts pathological vaginal alterations - a hallmark of GSM - in an established preclinical model of menopause.
Keywords: acellular biomaterial; extracellular matrix; hydrogel; menopause; vaginal atrophy.
© 2025 The Author(s). Advanced Materials published by Wiley‐VCH GmbH.
Conflict of interest statement
Dr. Christman is a co‐founder, consultant, and board member for, and holds equity interest and receives income from Ventrix Bio, Inc.
Figures



References
-
- Bachmann G., Santen R. J., (Ed: Chakrabarti R. L. B. A.), in UpToDate, Wolters Kluwer 2025.
-
- Hill K., Maturitas 1996, 23, 113. - PubMed
-
- Peacock K., Carlson K., Ketvertis K. M., in StatPearls, StatPearls Publishing,2023.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

