Clinical impact of diverting ileostomy and high-output stoma on adjuvant chemotherapy for rectal cancer: a retrospective cohort study
- PMID: 40751738
- PMCID: PMC12317863
- DOI: 10.1007/s00384-025-04971-1
Clinical impact of diverting ileostomy and high-output stoma on adjuvant chemotherapy for rectal cancer: a retrospective cohort study
Abstract
Purpose: Diverting ileostomy (DI) may cause fluid and electrolyte loss, potentially impairing the tolerability of adjuvant chemotherapy (ACT) in patients with rectal cancer. However, its clinical impact, especially in the presence of high-output stoma (HOS), remains unclear. This study aimed to evaluate the effects of DI and perioperative HOS on chemotherapy completion, dose intensity, and the incidence of severe adverse events (AEs).
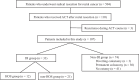
Methods: We retrospectively analyzed 107 patients with rectal cancer who underwent curative resection and received postoperative ACT between June 2012 and December 2024 at Tottori University. Chemotherapy completion, relative dose intensity (RDI), and grade ≥ 3 AEs were compared between patients with and without DI. A subgroup analysis assessed the influence of HOS among DI patients.
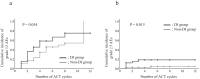
Results: Chemotherapy completion rate and RDI were comparable between patients with and without DI. However, the incidence of grade ≥ 3 AEs was significantly higher in the DI group than in the non-DI group (18.2% vs. 4.1%, P = 0.015), and DI was identified as an independent risk factor in multivariate analysis (odds ratio [OR] 5.749, P = 0.022). Among patients with DI, those with HOS had a significantly lower oxaliplatin RDI than those without HOS (37.5% vs. 75.0%, P = 0.007), and HOS independently predicted failure to complete oxaliplatin-based regimens (OR 13.423, P = 0.039).
Conclusions: While DI does not impair overall chemotherapy delivery, it is associated with increased early toxicity. HOS may compromise oxaliplatin administration and should prompt early recognition and targeted supportive interventions.
Keywords: Adjuvant chemotherapy; Diverting ileostomy; High-output stoma; Rectal cancer.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethical approval: This study was performed in line with the principles of the Declaration of Helsinki. The present study was approved by the Certified Review Board of Tottori University Hospital (approval number: 21A075). Ethics approval and consent to participate: All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional review board of ethics committee and national research committee with the 1964 Helsinki declaration and its later amendments. Disclosure: Authors declare no Conflict of Interests for this article. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Impact of the Diverting Stoma on Renal Function.Dis Colon Rectum. 2024 Dec 1;67(12):1576-1583. doi: 10.1097/DCR.0000000000003517. Epub 2024 Sep 11. Dis Colon Rectum. 2024. PMID: 39260442
-
Postoperative adjuvant chemotherapy in rectal cancer operated for cure.Cochrane Database Syst Rev. 2012 Mar 14;2012(3):CD004078. doi: 10.1002/14651858.CD004078.pub2. Cochrane Database Syst Rev. 2012. PMID: 22419291 Free PMC article.
-
Complications associated with loop ileostomy: analysis of risk factors.Tech Coloproctol. 2024 May 27;28(1):60. doi: 10.1007/s10151-024-02926-2. Tech Coloproctol. 2024. PMID: 38801595
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Hysterectomy with radiotherapy or chemotherapy or both for women with locally advanced cervical cancer.Cochrane Database Syst Rev. 2015 Apr 7;(4):CD010260. doi: 10.1002/14651858.CD010260.pub2. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2022 Aug 22;8:CD010260. doi: 10.1002/14651858.CD010260.pub3. PMID: 25847525 Updated.
References
-
- Bray F, Laversanne M, Sung H et al (2024) Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 74:229–263. 10.3322/caac.21834 - PubMed
-
- Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB (2019) Colorectal cancer. Lancet 394:1467–1480. 10.1016/s0140-6736(19)32319-0 - PubMed
-
- Benson AB, Venook AP, Adam M et al (2024) NCCN guidelines® insights: rectal cancer, version 3.2024. J Natl Compr Canc Netw 22:366–375. 10.6004/jnccn.2024.0041 - PubMed
-
- Kuebler JP, Wieand HS, O’Connell MJ et al (2007) Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol 25:2198–2204. 10.1200/jco.2006.08.2974 - PubMed
-
- André T, Boni C, Navarro M et al (2009) Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol 27:3109–3116. 10.1200/jco.2008.20.6771 - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

